Publications
Take a look at our recent peer-reviewed publications or book chapters!2024
![[85] Guidelines toward Reliable Facets Characterization for Structure-Sensitive Reactions](https://dusselier-lab.org/uploads/media/cache/default/uploads/4d1fbcda6f61218815a08ea0d4fe84d8.jpeg)
[85] Guidelines toward Reliable Facets Characterization for Structure-Sensitive Reactions 26-04-2024
Synthesizing defined facets/morphologies with atomic precise arrangements and testing them to control catalytic activity and selectivity is gaining traction. Researchers from different backgrounds took serious initiatives to explore this young area of research. Since there are no clear guidelines in the literature demonstrating the ideal method (or a method that most approximates reality) in determining the facets of the materials, it can be seen that there are many recent studies which proposed methods that are highly prone to overinterpretation or even wrong, which leads to precarious conclusions in this field. Therefore, it is our aim here to demonstrate the main points of confusion between the definitions and practices. Our contribution concludes with recommendations and an outlook.
![[84] Exploring Cage Effects of Alkane Adsorption on SSZ-39 (AEI) and SSZ-13 (CHA) Zeolites: A Comparative Study](https://dusselier-lab.org/uploads/media/cache/default/uploads/eff407faf3d2a909633e71b05004b470.jpeg)
[84] Exploring Cage Effects of Alkane Adsorption on SSZ-39 (AEI) and SSZ-13 (CHA) Zeolites: A Comparative Study 19-04-2024
Despite the recognized importance of the unique basket shape of the AEI zeolite cage in catalytic and separative applications, fundamental research on the adsorption properties of SSZ-39 (AEI-type) is still missing. Here, the adsorption properties of n-alkanes on a Na-SSZ-39 zeolite up to 320 °C were investigated by using inverse gas chromatography (IGC). Our findings reveal an unusual nonmonotonic increase of the Henry constant with increasing carbon number, marked by local minimum, signifying the presence of so-called cage effects. These cage effects are a direct result of the combination of small pores connected by larger cages found in the AEI framework and have only been experimentally reported for the CHA topology. A coiled-chain adsorption mechanism has been proposed using thermodynamic analysis, where long n-alkanes undergo a transition from one- to two-cage adsorption. A comparison with Na-SSZ-13 (CHA-type), a zeolite with a comparable cage-window structure and silica–alumina ratio, revealed that the local minimum of Henry constant occurs at n-octane for Na-SSZ-39 and n-nonane for Na-SSZ-13, even though both samples have comparable cage dimensions. Additionally, our results were compared to all available experimental data in the existing literature on cage effects in small-pore zeolites. Comparison of low-, medium-, and high silica CHA-type zeolites highlighted that Si/Al-ratios and cations impact the adsorption mechanism and the magnitude of cage effects for n-alkanes. Our findings open the door to synthetic attempts to actively influence and design adsorbents with tailored local minima.
![[83] Solvent-driven isomerization of muconates in DMSO: reaction mechanism and process sustainability](https://dusselier-lab.org/uploads/media/cache/default/uploads/a7ee94cd3d514d5f6e696c6c6db8b9e2.gif)
[83] Solvent-driven isomerization of muconates in DMSO: reaction mechanism and process sustainability 03-04-2024
The production of renewable chemicals and monomers is fundamental for transitioning to a future circular economy. Currently, cis,cis-muconic acid (ccMA) is a bio-sourced platform chemical with great potential for added-value chemicals, monomers, and specialty polymers. Among the three isomers, the trans,trans (tt-isomer) stands out due to its reactivity for polymerization and unique ability as a substrate for the Diels–Alder cycloaddition reaction. Whereas earlier research has focused on producing this isomer, the most promising solvent-driven isomerization in DMSO-containing water yields moderate ttMA due to a competitive ring-closing lactonization reaction, especially in highly concentrated systems. This work highlights the unique ability of DMSO, among several other solvents, to produce ttMA. In addition, we report the effect of the acidity of the initial MA concentration and the amount of water on the lactonization reaction. Control of reaction conditions and use of muconates (diethyl muconates = DEM) countered the competitive lactonization, reaching >90% tt-isomer selectivity. The involvement of water and DMSO in the isomerization mechanism was investigated in detail by probing the reaction mechanism with in situ NMR. Identifying the reaction products and several intermediates led us to propose a plausible mechanism. Based on this knowledge, condition optimization led to a significant thirty-fold ttDEM productivity improvement, viz. from 10 to 328 mM h−1. The DEM can be isolated almost quantitatively from the DMSO solvent system by extraction
![[82] Methionine and its hydroxy analogues: the paths toward their sustainable chemical synthesis](https://dusselier-lab.org/uploads/media/cache/default/uploads/ed070d37a12f25ee5565e24560fcca14.gif)
[82] Methionine and its hydroxy analogues: the paths toward their sustainable chemical synthesis 22-02-2024
Methionine (Met) and its hydroxy analogue (MHA) are important components of a multimillion tonne scale commodity market of supplements used in human and livestock nutrition. Currently the industrial chemical synthesis of Met and MHA depends on petroleum-derived feedstock, such as propene. Additionally, the conventional synthetic methods involve dealing with highly toxic compounds, such as acrolein or cyanide. Substituting the conventional processes with new ones dependent on bio-based feedstocks, and involving safer chemistries, will be of great importance to reach a sustainable future. This review discusses the current chemical processes to synthesize Met and MHA and critically assesses different approaches aiming to improve the sustainability of its key feedstock, namely acrolein, or the overall synthesis. The strengths, weaknesses, future opportunities and threats for each synthetic approach are highlighted
![[81] Support effects in vanadium incipient wetness impregnation for oxidative and non-oxidative propane dehydrogenation catalysis](https://dusselier-lab.org/uploads/media/cache/default/uploads/1baf433c1e8c7331465a47c2bb8e5427.png)
[81] Support effects in vanadium incipient wetness impregnation for oxidative and non-oxidative propane dehydrogenation catalysis 28-01-2024
- Incipient Wetness impregnation: Delve into the details of traditional incipient wetness impregnation Vanadium.
- Superfical density comparisons: Our impregnation technique ensures equivalent vanadium superficial density comparison with different supports.
- Assessing and Visualizing Results: Introducing the innovative strategy of Wisher and box plots to represent our findings clearly and concisely.
- Comparative Excellence: CO2-assisted propane dehydrogenation compared with traditional methods, highlighting standout MCM-41 performance in high-pressure OPDH.
2023
![[80] Inverse gas chromatography study of n-alkane and 1-alkene adsorption on pure-silica LTA (ITQ-29) and CHA](https://dusselier-lab.org/uploads/media/cache/default/uploads/7f70063a22796389d1323ec8e6c41b6c.jpeg)
[80] Inverse gas chromatography study of n-alkane and 1-alkene adsorption on pure-silica LTA (ITQ-29) and CHA 30-11-2023
Inverse gas chromatography was used to investigate the Henry adsorption constants, adsorption enthalpies and entropies of n-alkanes and 1-alkenes up to C12 on the small-pore pure-silica zeolites LTA (ITQ-29) and CHA (Si-CHA). Small hydrocarbons with two adjacent single bonds (such as propane and 1-butene) are shown to experience strong mass transfer limitations due to the small pore window of these zeolites. In contrast, these mass transfer limitations were absent for hydrocarbons with alternating single- and double bonds (such as propylene and butadiene). The extent of this effect decreases with increasing carbon numbers on both zeolites. For Si-LTA, a typical monotonous increase in Henry constant with carbon number is observed for both n-alkanes and 1-alkenes. In contrast, a non-monotonous increase in Henry constants with carbon number, so-called cage (nestling) effect, is observed for SiCHA. This effect is observed experimentally for 1-alkenes for the first time. Overall, our study sheds light on remarkable mass transfer limitations and cage effects that affect n-alkanes and 1-alkenes adsorption in these small-pore zeolites.
![[79] From tetroses to methionine hydroxy analogues through Sn (IV) Lewis acid catalysis using methanethiol as sulphur feedstock](https://dusselier-lab.org/uploads/media/cache/default/uploads/6a384895dedc7a8b191b68bbe2be4f56.jpeg)
[79] From tetroses to methionine hydroxy analogues through Sn (IV) Lewis acid catalysis using methanethiol as sulphur feedstock 27-11-2023
Methionine hydroxy analogues (MHAs) form part of a million tonne scale commodity market of compounds useful in livestock nutrition. The chemocatalytic synthesis of MHAs from carbohydrates is an important albeit understudied transformation en route to a sustainable and circular economy. Here we report our first results for the conversion of tetroses to MHAs in the presence of methanethiol (MeSH) and SnCl4·5H2O. Our study allowed to corroborate the major reaction pathways involved in this Lewis acid-catalysed transformation. It was also found that the ratio between MeSH and MeOH, the pressure of inert gas, and the water content strongly affect the reaction performance. When these parameters were combined the yield and selectivity towards MHAs increased to the highest reported value of 38 % in one hour reactions at 413 K. Our findings have the potential to unlock the sustainable synthesis of MHAs from renewable feedstock such as tetroses and other carbohydrates.
![[78] Electro-Assisted Synthesis of Sn-Beta Zeolite Leads to Record Tin Incorporation and Superior Lewis Acid Catalysis](https://dusselier-lab.org/uploads/media/cache/default/uploads/5f987ca8f3b547ab8c1b97dee616a3ff.jpeg)
[78] Electro-Assisted Synthesis of Sn-Beta Zeolite Leads to Record Tin Incorporation and Superior Lewis Acid Catalysis 06-11-2023
Tin-incorporated zeolites present a recognizable class of microporous materials widely used
to handle biomass valorization and selective oxidation challenges. Sn-BEA is among the most
catalytically valuable metallosilicate. However, in bottom-up synthesis, incorporation of the
metal is hampered, narrowing Lewis acid site density and, therefore, catalyst productivity.
Here, we present a novel strategy for hydrothermal preparation of Sn-BEA zeolite in hydroxide
media, employing our new Electro-Assisted Synthesis technique. Based on the controlled
anodic release of Sn from a metal electrode in a crystallizing siliceous zeolite mixture, we can
design tin-rich zeolites with record Si/Sn ratios as low as 14. The resulting Lewis acid
characteristics are unparalleled for bottom-up Sn-BEA and the small crystals are of benefit in
catalysis. One demonstration here in catalysis showed a three-fold increased performance (per
g of zeolite) versus a classic fluoride-made material in triose sugar conversion. We hypothesize
that both studied zeolites suffer from different types of mass transport limitations
![[77] Renewable and safer bisphenol A substitutes enabled by selective zeolite alkylation](https://dusselier-lab.org/uploads/media/cache/default/uploads/b45401983e47770a553f8c0bddbbcca0.jpeg)
[77] Renewable and safer bisphenol A substitutes enabled by selective zeolite alkylation 02-11-2023
Bisphenol A (BPA) is an industrial chemical that has been used for the manufacturing of polycarbonate plastics, epoxy resins and other consumer products, including food contact materials and dental resins. However, its petrochemical origin and adverse health effects, such as xeno-oestrogenic activity (EA), pose a challenge for sustainability. Here we show a green synthetic pathway towards genuinely sustainable BPA alternatives from a renewable lignin source. At the heart of this production route is a selective alkylation reaction between bio-based arene (for example, guaiacol) and alkene (for example, isoeugenol) molecules, catalysed by a recyclable zeolite catalyst H-USY. Zeolite catalysis favours regioselectivity and prompts higher reaction rate and chemoselectivity, enabling high yields of bisguaiacols. The synthesized bisguaiacols can be valorized into high-molecular-weight thermoplastic and thermosetting polymers with promising thermophysical properties, showcasing their potential as BPA replacements. This work progresses beyond the state of the art by demonstrating that renewability is not only a goal but also a means for safer chemicals.
![[76] Engineering Lewis Acidity in Zeolite Catalysts by Electrochemical Release of Heteroatoms during Synthesis](https://dusselier-lab.org/uploads/media/cache/default/uploads/a1907ba557a2fffca7e96219f2b6aed0.jpeg)
[76] Engineering Lewis Acidity in Zeolite Catalysts by Electrochemical Release of Heteroatoms during Synthesis 02-06-2023
The creation of heteroatom nodes in zeolite frameworks is a challenging but rewarding pathway to superior materials for numerous catalytic applications. Here, we present a novel method for precise control over heteroatom incorporation by in situ anodic release of a desired metal during hydrothermal zeolite synthesis. The generic character of the technique and the applicability of the new synthesis reactor are shown across 3 zeolite structures crystallized and 4 electrode metals in two pH zones and by offering access to a new mixed-metal zeolite. The timed and voltage-controlled metal release offers a minimized interference between the metal precursor state and critical events in the zeolite’s crystallization. A mechanistic study for Sn-MFI revealed the key importance of controlled release: while keeping its concentration lower than in batch, a lot more Sn can be incorporated into the framework. The method grants access to 10× increased framework Lewis acid site densities (vs batch controls) for the most relevant stannosilicates. As a proof, the electro-made materials demonstrate higher productivity than their classic counterparts in lactate catalysis. This innovative approach effectively expands the synthesis space of zeolites.
![[75] Sustainable polythioesters via thio(no)lactones: monomer synthesis, ring-opening polymerization, end-of-life considerations and industrial perspectives](https://dusselier-lab.org/uploads/media/cache/default/uploads/ca62e5d22c74ab82ba7f415a4fd8ffdb.png)
[75] Sustainable polythioesters via thio(no)lactones: monomer synthesis, ring-opening polymerization, end-of-life considerations and industrial perspectives 17-01-2023
As the environmental effects of plastics are of ever greater concern, the industry is driven towards more sustainable polymers. Besides sustainability, our fast-developing society imposes the need for highly versatile materials. While aliphatic polyesters (PEs) are widely adopted and studied as next-generation biobased and (bio)degradable materials, their sulfur-containing analogs, polythioesters (PTEs), only recently gained attention. Nevertheless, the introduction of S-atoms is known to often enhance thermal, mechanical, electrochemical, and optical properties, offering prospects for broad applicability. Furthermore, thanks to their thioester-based backbone, PTEs are inherently susceptible to degradation, giving them a high sustainability potential. The key route to PTEs is via ring-opening polymerization (ROP) of thio(no)lactones. This review critically discusses the (potential) sustainability of the most relevant state-of-the-art in every step from sulfur source to end-of-life treatment options of PTEs, obtained via ROP of thio(no)lactones. The benefits and drawbacks of PTEs versus PEs are highlighted, including their industrial perspective.
2022
![[74] Synthesis strategies to control the Al distribution in zeolites: thermodynamic and kinetic aspects](https://dusselier-lab.org/uploads/media/cache/default/uploads/6508f36a14ee44730c8945c8a251ff1b.jpeg)
[74] Synthesis strategies to control the Al distribution in zeolites: thermodynamic and kinetic aspects 23-12-2022
The activity and selectivity of acid-catalyzed chemistry is highly dependent on the Brønsted and Lewis acid sites generated by Al substitutions in a zeolite framework with the desired pore architecture. The siting of two Al atoms in close proximity in the framework of high-silica zeolites can also play a decisive role in improving the performance of redox catalysts by producing exchangeable positions for extra-framework multivalent cations. Thus, considerable attention has been devoted to controlling the Al incorporation through direct synthesis approaches and post-synthesis treatments to optimize the performance as (industrial) solid catalysts and to develop new acid- and redox-catalyzed reactions. This Feature Article highlights bottom-up synthetic strategies to fine-tune the Al incorporation in zeolites, interpreted with respect to thermodynamic and kinetic aspects. They include (i) variation in extra-framework components in zeolite synthesis, (ii) isomorphous substitution of other heteroatoms in the zeolite framework, and (iii) control over the (alumino)silicate network in the initial synthesis mixture via in-situ and ex-situ methods. Most synthetic approaches introduced here tentatively showed that the energy barriers associated with Al incorporation in zeolites can be variable during zeolite crystallization processes, occurring in complex media with multiple chemical interactions. Although the generic interpretation of each strategy and underlying crystallization mechanism remains largely unknown (and often limited to a specific framework), this review will provide guidance on more efficient methods to prepare fine-tuned zeolites with desired chemical properties.
![[73] Steering Interzeolite Conversion with Alkali Metal Cations: Lithium Maximizes Al Proximity in SSZ-13 Zeolite Genesis](https://dusselier-lab.org/uploads/media/cache/default/uploads/55ddf2cfb0624af253063fe456e294d3.jpeg)
[73] Steering Interzeolite Conversion with Alkali Metal Cations: Lithium Maximizes Al Proximity in SSZ-13 Zeolite Genesis 23-12-2022
Zeolites have long been regarded as difficult to modify during synthesis, as their synthesis is governed by kinetic processes. Recent breakthroughs have made it possible to exert a certain degree of control over zeolite properties with more performant materials as a result. Here, we investigate the effects alkali cations have on high-silica FAU-to-CHA interzeolite conversion (IZC) and on the resulting aluminum distributions. In this way, by using Li-cations in conjunction with an organic structure directing agent, the first route to a “fully paired” (divalent cation capacity, Co2+/Al = 0.48) high-silica SSZ-13 zeolite is demonstrated. Lithium shows great potential in steering IZC synthesis as it speeds up crystallization, and evidence was gathered in favor of a more elaborate mechanism of IZC in which dissolved Al-rich oligomers crash out of solution first and possibly spark nucleation. These findings help in gaining insights into a more general theory on zeolite nucleation in heterogeneous environments such as IZC. Furthermore, the “fully paired” sample has great potential for ion-exchanged zeolite catalysis or in adsorbents.
![[72] Truly combining the advantages of polymeric and zeolite membranes for gas separations](https://dusselier-lab.org/uploads/media/cache/default/uploads/4bddef1adf550685b87adefb41365c91.png)
[72] Truly combining the advantages of polymeric and zeolite membranes for gas separations 16-12-2022
Mixed-matrix membranes (MMMs) have been investigated to render energy-intensive separations more efficiently by combining the selectivity and permeability performance, robustness, and nonaging properties of the filler with the easy processing, handling, and scaling up of the polymer. However, truly combining all in one single material has proven very challenging. In this work, we filled a commercial polyimide with ultrahigh loadings of a high–aspect ratio, CO2-philic Na-SSZ-39 zeolite with a three-dimensional channel system that precisely separates gas molecules. By carefully designing both zeolite and MMM synthesis, we created a gas-percolation highway across a flexible and aging-resistant (more than 1 year) membrane. The combination of a CO2-CH4 mixed-gas selectivity of ~423 and a CO2 permeability of ~8300 Barrer outperformed all existing polymer-based membranes and even most zeolite-only membranes.
![[71] Brønsted acid catalysis opens a new route to polythiolesters via the direct condensation of thiolactic acid to thiolactide](https://dusselier-lab.org/uploads/media/cache/default/uploads/de569d33ee9de712d3b2cc7b65a40e10.jpeg)
[71] Brønsted acid catalysis opens a new route to polythiolesters via the direct condensation of thiolactic acid to thiolactide 12-12-2022
Polythiolesters (PTEs) are a rather underinvestigated group of sulfur-containing polymers with high potential as sustainable materials due to their enhanced de-polymerizability under specific conditions compared to polyesters (PEs). Poly(thiolactic acid), the sulfur analog of poly(lactic acid) (PLA), one of the most prominent biobased and biodegradable PEs, was only very recently obtained with relatively high molecular weight by the ring-opening polymerization of thiolactide (TLD). Here, we demonstrate a novel synthesis route to TLD by the direct condensation of thiolactic acid (TLA) with Brønsted acid catalysts in the absence of co-reagents or stoichiometric additives. Homogeneous sulfonic acid catalysts afforded a TLD yield of up to 80% directly from racemic TLA. Heterogenizing the catalytic system is challenging in which sulfonic acid containing ion-exchange resins enabled a maximum yield of 39%, while lower yields were obtained with Brønsted acid beta zeolites (H-BEA, up to 28%). Even though these zeolites are known to be very apt for the synthesis of lactide from lactic acid, there are profound differences due to the presence of sulfur. First, TLA showed a lower tendency toward (poly)condensation than LA, reducing the need for shape-selective catalysis. Second, very strong interactions of TLA, compared to LA, with the zeolite were seen by FT-IR and hypothesized to counteract catalytic activity and induce diffusion limitations or sulfur poisoning. The new methods were assessed according to the principles of green chemistry and compared to the other two existing methods in the literature, showing a significant improvement with regard to sustainability for our Brønsted acid catalyzed direct dehydration of TLA. Finally, the Brønsted acid derived TLD was successfully purified and polymerized into PTEs with molecular weights up to 56.0 kg mol−1, revealing some insights into the TLD ring-opening mechanism.
![[70] Fabrication of a sinter-resistant Fe-MFI zeolite dragonfruit-like catalyst for syngas to aromatics conversion](https://dusselier-lab.org/uploads/media/cache/default/uploads/2d8bfecec940e272a4cb24d46fad9acc.jpeg)
[70] Fabrication of a sinter-resistant Fe-MFI zeolite dragonfruit-like catalyst for syngas to aromatics conversion 28-10-2022
Direct conversion of syngas to aromatics has great potential to decrease fossil fuel dependence. Here, a unique structured hybrid catalyst composed of Fe3O4 nanoparticles intimately dispersed inside an acidic zeolite is developed. 1 to 4 nm sized Fe3O4 nanoparticles end up evenly dispersed in an acidic and slightly mesoporous Al-ZSM-5 based on Fe3O4 restructuring during co-hydrothermal synthesis using organosilane modification. A very high aromatic productivity of 214 mmolaromatics h-1 gFe-1 can be obtained with a remarkable 62% aromatic selectivity in hydrocarbons. This catalyst has excellent sintering resistance ability and maintains stable aromatics production over 570 h. The synthetic insights that postulate a mechanism for the metastable oxide-zeolite reorganization during hydrothermal synthesis could serve as a generic route to sinter-resistant oxide-zeolite composite materials with uniform, well-dispersed oxide nanoparticles in close intimacy with - and partially confined in - a zeolite matrix.
![[69] A Critical Revisit of Zeolites for CO2 Desorption in Primary Amine Solution Argues Its Genuine Catalytic Function](https://dusselier-lab.org/uploads/media/cache/default/uploads/a02c123b4341b523e728b0ad88b0f2a7.jpeg)
[69] A Critical Revisit of Zeolites for CO2 Desorption in Primary Amine Solution Argues Its Genuine Catalytic Function 14-09-2022
Zeolites are the most widely reported solid materials that are used in primary amine-containing postcombustion CO2 capture processes for quicker solvent regeneration at a lower energy consumption. Catalytic solvolysis of the carbamate intermediate, assisted by the Brönsted acid sites (BAS), is commonly accepted as an explanation. However, there is little, if any, attention given to the regeneration of BAS in such basic amine-rich solution. Herein, we revisit the role of zeolite for CO2 desorption in primary amine solution at room to moderately elevated temperature ranges. We noticed indeed an accelerating effect on the CO2 desorption rate in the presence of BAS. Both their numbers as well as their accessibility for the amine-CO2 adduct (i.e., carbamate) (direct) or amine (indirect pathway) are important. However, we also noticed, using spectroscopic techniques and by zeolite reuse, a very strong chemical interaction between BAS and the regenerated amine. This suggests that BAS recovery to close the catalytic cycle is difficult and that zeolites can hardly be considered as genuine catalysts, but rather, this study concludes a merely stoichiometric effect of the zeolites for the overall desorption process, and this is in contrast to reference oxides such as TiO(OH)2.
![[68] Toward Industrially Relevant Sn-BETA Zeolites: Synthesis, Activity, Stability, and Regeneration](https://dusselier-lab.org/uploads/media/cache/default/uploads/f8051120eb6a188827cdbe9327ecbd58.jpeg)
[68] Toward Industrially Relevant Sn-BETA Zeolites: Synthesis, Activity, Stability, and Regeneration 14-09-2022
Lewis acidic Snβ zeolites have emerged as highly interesting catalysts for a variety of biomass-related reactions. During the last decades, a vast body of research has focused on unravelling the identity of the (most) active Sn sites. Recent research has shifted its focus from fine-tuning one single type of active Sn-site toward optimizing the overall catalytic activity and stability of the Snβ zeolite, which is more suitable from an industrial point of view. After delving into a discussion of the Sn active sites, this Perspective highlights the recent developments that are essential for industrial implementation of Snβ zeolites, with an emphasis on the most recent insights and findings for improving the catalyst’s productivity and stability.
![[67] Reshaping the Role of CO2 in Propane Dehydrogenation: From Waste Gas to Platform Chemical](https://dusselier-lab.org/uploads/media/cache/default/uploads/73d0591289271d2b60b18d9d94102ffd.jpeg)
[67] Reshaping the Role of CO2 in Propane Dehydrogenation: From Waste Gas to Platform Chemical 18-07-2022
The valorization of CO2 appeals to the chemical industry due to the reduction in greenhouse gas emissions and the ability to offer more renewable products. Propylene production is the second largest process in the chemical industry, and it strongly depends on fossil fuel feedstocks. Coupling CO2 reduction with propane dehydrogenation boosts conversion and produces CO, a valuable platform chemical currently synthesized by fossil-methane reforming. In this work, (i) we demonstrate the environmental benefits of coupling CO2 with a life-cycle assessment under industrial conditions, potentially reducing emissions by 3 tCO2-eq per ton of propylene produced. (ii) We screen supported catalytic materials─both known and novel─with a focus on propane and CO2 reaction mechanisms under industrial reaction conditions of 400–700 °C and pressures of 1–6 barg (redox: V, Galinstan, In, Mo, Mn, Bi, Sb, Ta; nonredox: Cr, Ga, co, Al, Zn, Au, Zr, Ag, W, La, Cu; and some alloy combinations). We evaluate each material under the kinetic regime, and we quantify reaction, side reaction, and deactivation kinetics (coking, cracking, and dry-reforming), as well as the regeneration ability. We then classify them based on their dominant mechanisms (direct CO2 assistance or indirect with H2 via reverse water gas shift) and identify each catalyst’s strengths and weaknesses. Finally, (iii) we correlate our database of experimental results of 22 active metals/metal oxides with the Tamman temperature and density functional theory (DFT)-based oxygen vacancy formation energies. We discovered that oxygen mobility plays a crucial role in the kinetics of reoxidation with CO2 and the overall balance of active sites related to dehydrogenation and reoxidation.
![[66] Catalytic amination of lactic acid using Ru–zeolites](https://dusselier-lab.org/uploads/media/cache/default/uploads/5609c3d8d893fd6a85fd59ee58e6a19e.gif)
[66] Catalytic amination of lactic acid using Ru–zeolites 17-07-2022
In this work we investigate the synthesis of alanine from lactic acid, a biobased platform chemical, using ammonia as a nitrogen source and Ru/zeolite catalysts. We report a high alanine selectivity when using Ru/BEA of 80–93%. Reaction side products were identified as ethanol, propionic acid or propanamide and the reaction mechanism was investigated. We further optimised reaction conditions resulting in turn over numbers five times higher than previously reported and could reduce Ru leaching by 30–40%. However, leaching and catalyst stability remains a concern. Furthermore, we critically analyse the benefits of Ru/zeolites versus their stability under the basic, high temperature reaction conditions.

BOOK: Biodegradable Polymers in the Circular Plastics Economy 17-07-2022
As the lasting impact of humanity’s reliance on plastics comes into focus, scholars have begun to seek out solutions to plastic litter. In Biodegradable Polymers in the Circular Plastics Economy, an accomplished team of researchers delivers a focused guide (1) to understand plastic degradation and its role in waste hierarchy besides recycling, and (2) to create and use biodegradable plastics where appropriate. Created preferably from renewable resources, these eco-friendly polymers provide an opportunity to create sustainable and lasting solutions to the growing plastic-driven pollution problem.
The broad approach to this handbook allows the authors to cover all aspects of these emerging materials, ranging from the problems present in the current plastics cycle, to the differences in type, production, and chemistry available within these systems, to end-of-life via recycling or degradation, and to life-cycle assessments. It also delves into potential commercial and policy issues to be addressed to successfully deploy this technology.
Readers will also find:
- A thorough introduction to biodegradable polymers, focusing not only on the scientific aspects, but also addressing the larger political, commercial, and consumer concerns
- Mechanisms of biodegradation and the environmental impact of persistent polymers
- An in-depth discussion of degradable/hydrolysable polyesters, polysaccharides, lignin-based polymers, and vitrimers
- Management of plastic waste and life cycle assessment of bio-based plastics
Biodegradable Polymers in the Circular Plastics Economy is the perfect overview of this complicated but essential research field and will appeal to polymer chemists, environmental chemists, chemical engineers, and bioengineers in academia and industry. The book is intended as a step towards a circular plastics economy that relies heavily on degradable plastics to sustain it.
![[65] Establishing the Reaction Pathways of the Catalytic Conversion of Erythrulose to Sulphides of Alpha-Hydroxy Thioesters and Esters](https://dusselier-lab.org/uploads/media/cache/default/uploads/cd99b2b55284d2e1c4a2dfc19332ee63.jpeg)
[65] Establishing the Reaction Pathways of the Catalytic Conversion of Erythrulose to Sulphides of Alpha-Hydroxy Thioesters and Esters 17-07-2022
Sulphides of alpha-hydroxy thioesters and esters (SAH(T)Es) are important fine chemicals and have great potential as platform molecules. SAH(T)Es are typically synthesized from fossil sources while little is known regarding their synthesis from carbohydrates. We report our findings about the one-pot chemocatalytic conversion of erythrulose (ERU) to SAH(T)Es. Sn, Mo and W chlorides were the most selective catalysts towards the synthesis of S-butyl-4-butylthio-2-hydroxybutanethioate (BBTHBT) in 1-BuSH. The selectivity towards BBTHBT was impacted by the formation of different thioacetals (TAs). The addition of either KOH, H2O or MeOH was effective to decrease TAs formation and increase selectivity towards BBTHBT (or its ester). A kinetic profile, in situ 13C NMR measurements and experiments at different temperatures complement our studies to unravel the complicated reaction network involved in the conversion of ERU to SAH(T)Es. This insight provides a solid foundation for future improvements in the sustainable synthesis of SAH(T)Es.
2021
![[64] Tandem Reduction–Reoxidation Augments the Catalytic Activity of Sn-Beta Zeolites by Redispersion and Respeciation of SnO2 Clusters](https://dusselier-lab.org/uploads/media/cache/default/uploads/01c0307e4baf20888c56173b1aa9c429.gif)
[64] Tandem Reduction–Reoxidation Augments the Catalytic Activity of Sn-Beta Zeolites by Redispersion and Respeciation of SnO2 Clusters 01-12-2021
High dispersion of Sn in beta (β) zeolites is pivotal to obtain highly active Snβ catalysts with high productivity. However, at higher Sn loadings, Sn dispersion and the activity per Sn decrease. The present work highlights the augmentation of catalytic activity in the Baeyer–Villiger oxidation and Meerwein–Ponndorf–Verley reactions of the as-synthesized Snβ catalysts with Sn loadings of up to 10 wt % by tandem reduction–reoxidation and discusses the effect in terms of Lewis acid (LA) density, Sn dispersion, Sn speciation, and catalytic performance. To do so, nontreated, reduced, and reduced–reoxidized Snβ catalysts are characterized by N2 physisorption, X-ray diffraction (XRD), and a multitude of spectroscopic techniques such as temperature-programmed reduction-mass spectrometry (TPR-MS), probe Fourier transform infrared (FTIR), diffuse reflectance ultraviolet–visible (DRUV–vis), 119Sn Mössbauer, 119Sn magic angle spinning nuclear magnetic resonance (MAS NMR), inductively coupled plasma-atomic emission spectroscopy (ICP-AES), and X-ray photoelectron spectroscopy (XPS). Although not mutually exclusive, the higher catalytic activity seems to arise from different phenomena depending on the Sn loading. At a lower Sn content (<5 wt %), the higher activity arises from the redispersion of SnO2 clusters into smaller active Sn species, whereas at a higher Sn content (≥5 wt %), the catalytic activity seems to improve based on the respeciation of SnO2 clusters.
![[63] Engineering low-temperature ozone activation of zeolites: process specifics, possible mechanisms and hybrid activation methods](https://dusselier-lab.org/uploads/media/cache/default/uploads/c637d5c62c9f5f8503f5aeb1a81b4d44.jpeg)
[63] Engineering low-temperature ozone activation of zeolites: process specifics, possible mechanisms and hybrid activation methods 01-12-2021
Zeolite activation is the first, but often overlooked, post-synthetic treatment needed to transform an as-made material into an adsorbent or catalyst. Ozonation at low temperatures provides an alternative for conventional calcination treatments, with important consequences for zeolite stability and acidity. While sometimes used in the context of emptying novel less thermally stable structures, a thorough look at ozonation for bulk zeolites is missing. Decomposition of organic structure-directing agents was achieved for three OSDA-zeolite systems yielding empty SSZ-13, ZSM-5 and β zeolites at select ozonation conditions. The effect of various process parameters, as well as synthetic parameters, were investigated. Infrared and mass spectrometry after alternating O2-O3 activation mode experiments provide some mechanistic insights into ozonation. The rate-limiting steps of both low-temperature ozonation and air calcinations can be circumvented by low-temperature ozonation after medium-temperature calcination.
![[62] How Substituent Effects Influence the Thermodynamics and Kinetics of Gas-Phase Transesterification of Alkyl Lactates to Lactide using TiO2/SiO2](https://dusselier-lab.org/uploads/media/cache/default/uploads/2154b3ba52f60d0f25bd2d44ab55994c.jpeg)
[62] How Substituent Effects Influence the Thermodynamics and Kinetics of Gas-Phase Transesterification of Alkyl Lactates to Lactide using TiO2/SiO2 22-09-2021
Lactide, a crucial precursor of the bioplastic polylactic acid, was obtained through the gas-phase transesterification of alkyl lactates using a 5 wt% TiO2/SiO2 catalyst as an alternative to the conventional liquid-phase process. Lactide selectivity above 80% was achieved at 220 °C but conversion was thermodynamically limited at around 50% for all alkyl lactates. The nature of the ester alkyl chain had minimal impact of the thermodynamics of the reaction, but significantly influenced its kinetics. The Taft equation indicated that this observation was due to the polar effects of the alkyl chain. Mechanistic studies indicated lactide formed via a Langmuir-Hinshelwood mechanism between two lactate molecules. The derived kinetic expression was fitted to experimental data of methyl lactate transesterification, resulting in a reduced chi-squared of 0.99 and an activation energy of 89.8 kJ·mol-1. Initial rate kinetics confirmed that the proposed mechanism was valid for other alkyl lactate species.
![[61] The selective formation of α-Fe(II) sites on Fe-zeolites through one-pot synthesis](https://dusselier-lab.org/uploads/media/cache/default/uploads/aff3b098a819c9540d44f55188fecefe.jpeg)
[61] The selective formation of α-Fe(II) sites on Fe-zeolites through one-pot synthesis 10-09-2021
α-Fe(II) active sites in iron zeolites catalyze N2O decomposition and form highly reactive α-O that selectively oxidizes unreactive hydrocarbons, such as methane. How these α-Fe(II) sites are formed remains unclear. Here different methods of iron introduction into zeolites are compared to derive the limiting factors of Fe speciation to α-Fe(II). Postsynthetic iron introduction procedures on small pore zeolites suffer from limited iron diffusion and dispersion leading to iron oxides. In contrast, by introducing Fe(III) in the hydrothermal synthesis mixture of the zeolite (one-pot synthesis) and the right treatment, crystalline CHA can be prepared with >1.6 wt % Fe, of which >70% is α-Fe(II). The effect of iron on the crystallization is investigated, and the intermediate Fe species are tracked using UV–vis-NIR, FT-IR, and Mössbauer spectroscopy. These data are supplemented with online mass spectrometry in each step, with reactivity tests in α-O formation and with methanol yields in stoichiometric methane activation at room temperature and pressure. We recover up to 134 μmol methanol per gram in a single cycle through H2O/CH3CN extraction and 183 μmol/g through steam desorption, a record yield for iron zeolites. A general scheme is proposed for iron speciation in zeolites through the steps of drying, calcination, and activation. The formation of two cohorts of α-Fe(II) is discovered, one before and one after high temperature activation. We propose the latter cohort depends on the reshuffling of aluminum in the zeolite lattice to accommodate thermodynamically favored α-Fe(II).
![[60] A Cooperative OSDA Blueprint for Highly Siliceous Faujasite Zeolite Catalysts with Enhanced Acidity Accessibility](https://dusselier-lab.org/uploads/media/cache/default/uploads/d1e402f8361680a2eb026508f2b7eb86.jpeg)
[60] A Cooperative OSDA Blueprint for Highly Siliceous Faujasite Zeolite Catalysts with Enhanced Acidity Accessibility 01-09-2021
A cooperative OSDA strategy is demonstrated, leading to novel high-silica FAU zeolites with a large potential for disruptive acid catalysis. In bottom-up synthesis, the symbiosis of choline ion (Ch+) and 15-crown-5 (CE) was evidenced, in a form of full occupation of the sodalite (sod) cages with the trans Ch+ conformer, induced by the CE presence. CE itself occupied the supercages along with additional gauche Ch+, but in synthesis without CE, no trans was found. The cooperation, and thus the fraction of trans Ch+, was closely related to the Si/Al ratio, a key measure for FAU stability and acidity. As such, a bottom-up handle for lowering the Al-content of FAU and tuning its acid site distribution is shown. A mechanistic study demonstrated that forming sod cages with trans Ch+ is key to the nucleation of high-silica FAU zeolites. The materials showed superior performances to commercial FAU zeolites and those synthesized without cooperation, in the catalytic degradation of polyethylene.
![[59] On the key role of aluminium and other heteroatoms during interzeolite conversion synthesis](https://dusselier-lab.org/uploads/media/cache/default/uploads/ec3f6057ce2b81f1f71f79a4696b61ac.png)
[59] On the key role of aluminium and other heteroatoms during interzeolite conversion synthesis 03-08-2021
(EMERGING INVESTIGATOR SERIES)
Interzeolite conversion, a synthesis technique for several zeolite frameworks, has recently yielded a large amount of high-performing catalytic zeolites. Yet, the mechanisms behind the success of interzeolite conversion remain unknown. Conventionally, small oligomers with structural similarity between the parent and daughter zeolites have been proposed, despite the fact these have never been observed experimentally. Moreover, recent synthesis examples contradict the theory that structural similarity between the parent and daughter zeolites enhances interzeolite conversion. In this perspective it is proposed that heteroatoms, such as aluminium, are key players in the processes that determine the successful conversion of the parent zeolite. The role of Al during parent dissolution, and all consecutive stages of crystallization, are discussed by revising a vast body of literature. By better understanding the role of Al during interzeolite conversions, it is possible to elucidate some generic features and to propose some synthetic guidelines for making advantageous catalytic zeolites. The latter analysis was also expanded to the interconversion of zeotype materials where heteroatoms such as tin are present.
![[58] Highly Dispersed Sn-beta Zeolites as Active Catalysts for Baeyer–Villiger Oxidation: The Role of Mobile, In Situ Sn(II)O Species in Solid-State Stannation](https://dusselier-lab.org/uploads/media/cache/default/uploads/ea48467063599940c4e4a058d3228cf5.gif)
[58] Highly Dispersed Sn-beta Zeolites as Active Catalysts for Baeyer–Villiger Oxidation: The Role of Mobile, In Situ Sn(II)O Species in Solid-State Stannation 04-02-2021
Solid-state incorporation of Sn into beta (β) zeolites is a fast and efficient method to obtain Lewis acidic Snβ catalysts with high activity. The present work emphasizes the fundamental role of the heat-treatment atmosphere in the solid-state incorporation of active Sn in zeolites. Via an array of characterization tools including N2-physisorption, X-ray diffraction, diffuse reflectance UV–vis spectrocopy, Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy, and 119Sn Mössbauer spectroscopy, it is shown that preheating under an inert atmosphere (pre-pyrolysis) prior to air-calcination affords Sn-β catalysts with the highest Sn dispersion and significantly less extra-framework SnO2 compared to the classic calcination. In situ characterization during pre-pyrolysis by temperature-programed decomposition–mass spectrometry, thermogravimetric analysis, and 119Sn Mössbauer spectroscopy reveals the in situ generation of Sn(II)O species that are more mobile than Sn(IV)O2 species generated during calcination. This mobility property essentially enables the high Sn dispersion in Snβ. Based on this knowledge, active sites per catalyst weight are maximized while retaining high turn-over frequencies for the Baeyer–Villiger oxidation reaction (300 h–1 at 80 °C). For Lewis acid densities above 200 μmol·g–1, the catalytic activity unexpectedly leveled off to 93 mM·h–1, even under kinetic control. We tentatively ascribe the activity plateau to the incorporation of Sn in less favorable T-sites at high Sn-loadings.
![[57] Boosting PLA melt strength by controlling the chirality of co-monomer incorporation](https://dusselier-lab.org/uploads/media/cache/default/uploads/467b5ca0dd81cb3edbc90cd28b9c19c2.png)
[57] Boosting PLA melt strength by controlling the chirality of co-monomer incorporation 04-02-2021
Bio-based and degradable polymers such as poly(lactic acid) (PLA), have become prominent. In spite of encouraging features, PLA has a low melt strength and melt elasticity, resulting in processing and application limitations, that diminish its substitution potential vis-a-vis classic plastics. Here, we demonstrate a large increase in zero shear viscosity, melt elasticity, elongational viscosity and melt strength by random co-polymerization of lactide with small amounts, viz. 0.4-10 mol%, of diethylglycolide of opposite chiral nature. These enantiomerically pure monomers can be synthesized using one-step zeolite catalysis. Screening of the ester linkages in the final PLA chains by the ethyl side groups is suggested to create an expanding effect on the polymer coils in molten state by weakening of chain-chain interactions. This effect is suspected to increase the radius of gyration, enabling more chain entanglements and consequently increasing melt strength. A stronger melt could enable access to more cost-competitive and sustainable PLA-based biomaterials with a broader application window. Amongst others, blow molding of bottles, film blowing, fibre spinning and foaming could be facilitated by PLA materials exhibiting a stronger melt.
![[56] Interzeolite Conversion and the Role of Aluminum: Toward Generic Principles of Acid Site Genesis and Distributions in ZSM-5 and SSZ-13](https://dusselier-lab.org/uploads/media/cache/default/uploads/06f1766e469b319bdecfad61f4560453.gif)
[56] Interzeolite Conversion and the Role of Aluminum: Toward Generic Principles of Acid Site Genesis and Distributions in ZSM-5 and SSZ-13 04-02-2021
The performance of zeolite catalysts depends not only on the strength and number of Brønsted acid (or exchange) sites but also on synergistic effects derived from their proximity, in particular, and their distribution, in general. Little is known on the genesis of acid sites and site distributions in hydrothermal zeolite synthesis. By an extensive study of five crystallization systems yielding ZSM-5 (MFI) and SSZ-13 (CHA), with a focus on interzeolite conversion (IZC) methods, several synthesis factors and mechanisms that are key in determining the output acid site distribution have been identified. Key in this study were temporal synthesis profiles while probing the distribution and evolution of proximal acid sites with divalent cation capacity measurements. Over the course of different crystallizations, changing local charge distributions are detected, notably within crystalline materials upon prolonged exposure (maturation). Aluminum is clearly the key driver in IZC syntheses, from charge, dissolution, concentration, and mobility points of view. Quasigeneric principles for IZC syntheses are proposed, distinguishing between Al-loving and Al-averse systems, enabling a new degree of control over the acidity and ion-exchange properties of zeolites, of use to tailoring catalytic activity.
2020
![[55] Syntheses and chemical transformations of glycolide and lactide as monomers for biodegradable polymers](https://dusselier-lab.org/uploads/media/cache/default/uploads/2f95650dfd76f4e660598ce508c73c64.jpeg)
[55] Syntheses and chemical transformations of glycolide and lactide as monomers for biodegradable polymers 27-11-2020
Glycolide and lactide function as the commonly used diester monomers for the preparation of high-molecular weight, degradation-prone (co-)polyesters. Both glycolide and lactide-based polymers are widely used in medicine, pharmaceuticals, the food industry, and additive technologies. This review on the diesters, spanning research from 1833-1854 to the present, encompassing their structural peculiarities, physico-chemical properties, and the range of different methods for obtaining them – including reaction mechanisms – from lactic and glycolic acids, their esters, and halogen derivatives. The review also discusses the chemical transformations of lactide and glycolide (apart from ring-opening polymerization) into valuable organic and high-molecular compounds, such as acrylic acid, nitrogen-containing heterocycles, and functional polymers with novel properties.
![[54] MOF‐derived/zeolite hybrid catalyst for the production of light olefins from CO2](https://dusselier-lab.org/uploads/media/cache/default/uploads/b86414895ec7bf4998f89598691f66a4.jpeg)
[54] MOF‐derived/zeolite hybrid catalyst for the production of light olefins from CO2 12-08-2020
In this contribution we propose an alternative catalytic system based on MOF derivatives and small pore zeolites for the selective conversion of CO 2 into light olefins, using the lowest metal loadings and highest GHSV reported in literature. The catalyst synthesis involves deriving In‐Zr oxides from MOFs containing these metals in their structure, i.e. (Zr)UiO‐67‐bipy‐In, via direct calcination in the presence of the zeolite, avoiding co‐precipitation, washing and mixing steps. This effectively creates a truly bifunctional In‐Zr zeolite catalyst, opposed to physical mixtures of two catalysts using different precursors. The good dispersion and low loadings of the MOF‐ derived In‐Zr oxide supplemented with the strong acidity of chabazite‐type zeolites allows to couple the activation of CO 2 with C‐C coupling, obtaining space time yields of 0.1 mol of CO 2 converted to light olefins per gram of In per hour at 375ºC, under the GHSV conditions employed.
![[53] Zeolite Synthesis under Nonconventional Conditions: Reagents, Reactors, and Modi Operandi](https://dusselier-lab.org/uploads/media/cache/default/uploads/7dc7093a635fbccef97d2e7816091a2f.jpeg)
[53] Zeolite Synthesis under Nonconventional Conditions: Reagents, Reactors, and Modi Operandi 06-05-2020
A myriad of tetrahedral molecular sieve frameworks, often siliceous, can be calculated in silico. Only a tiny fraction (<0.1%) of these can be synthesized on purpose. Only a small fraction of these available frameworks, mostly those composed of only Si and Al as T-atoms, i.e. true zeolites, are used commercially. A gap thus exists between what should be possible (thermodynamically) and what can be produced (kinetically) and used in real life. Even if a synthesis is successful (in industry or academia), flexibility with regard to synthesis parameters - in terms of time, amount of unit operations, OSDA-efficiency, etc. - as well as the obtained material properties - in terms of Si/Al ratio, Al-distribution, T-atom variety, crystal size, etc. - remains limited. These limitations are not surprising since convention-al zeolite syntheses, i.e. hydrothermal synthesis in batch from amorphous or soluble Si- and Al-sources, have limited degrees of freedom (DOF). Typically, the type of ingredients, their ratios, a constant temperature, synthesis time and the absence or presence of agitation are varied. In order to take new steps towards more cost-competitive syntheses, and more importantly, zeolites with a greater flexibility in terms of structural properties, this review highlights all DOFs that can be introduced in addition to or on top of the conventional way of synthesis. By doing this, a distinction is made between non-conventional DOFs that influence the chemistry of the system (e.g. interzeolite conversion, charge density mismatch approach, ionothermal or free-radical assisted synthesis) and non-conventional DOFs that influence the physical environment (e.g. ultrasounds, alternative energy via microwaves or continuous set-ups). The review concludes with learnings, practical insights and future opportunities. In other words: which zeolite synthesis strategies really make a difference and which ones are just tweaking around the edges?
![[52] The role of pretreatment in the catalytic valorization of cellulose](https://dusselier-lab.org/uploads/media/cache/default/uploads/0630b6c79a40ff9ba7c2cb57a96a7048.jpeg)
[52] The role of pretreatment in the catalytic valorization of cellulose 20-03-2020
The use of fossil resources with the concerning climate change has pushed the development of alternative and sustainable feedstocks for energy, chemicals, and materials production. Lignocellulose is one of the most abundant and promising renewable carbon sources for the transition of a petroleum-based to a bio-based economy. Unlike liquid petroleum oil, the inherent complex polymer structure of the solid lignocellulose makes it recalcitrant for (selective) valorization. Hence, physical and/or chemical pretreatments are usually required to improve the reactivity and/or purity of the feedstock. Promising biorefinery concepts should be able to utilize all components of the lignocellulose. Since cellulose is the most abundant fraction in lignocellulose, this review emphasizes the progress and importance of pretreatment in the conversion of cellulose pulp into chemicals and fuels. The structural and chemical features such as crystallinity, particle size, porosity, and degree of polymerization that influence the reactivity of cellulose are overviewed whenever possible for different reaction types. The advantage and disadvantage of the different approaches for their determination are discussed in detail. The benefits of different pretreatments in bio-chemical and more particularly in chemo-catalytic conversion of cellulose are overviewed. Finally, the future and perspective of (ligno)cellulose pretreatment with regard to cellulose utilization are discussed.
![[51] Muconic acid isomers as platform chemicals and monomers in the biobased economy](https://dusselier-lab.org/uploads/media/cache/default/uploads/8f73ad37fa1e8267e9b703783de5d850.jpeg)
[51] Muconic acid isomers as platform chemicals and monomers in the biobased economy 17-02-2020
Muconic acid (MA) is a high value-added dicarboxylic acid with conjugated double bonds, presenting three isomeric forms, i.e., cis,cis-MA, cis,trans-MA and trans,trans-MA. Its production is garnering increased interest owing to its potential as a starting material for the synthesis of value-added products as well as by being a versatile monomer for the production of specialty polymers. This review presents a systematic overview of production and synthesis routes to MA isomers as well as the routes for its final valorization. The production of MA through chemical pathways and the progress developed in the biotechnological pathways will be discussed. Traditional and new processes for achieving successful isomerization into the value-added trans,trans form of MA are also discussed and compared, with their constraints and possible solutions. The valorization of the three different isomers of MA into value-added chemicals such as adipic or terephthalic acids and MA polymers are summarized. Finally, the review concludes with a thorough summary, a practical insights section and an outlook. This work thus offers new perspectives and guidelines to tackle the challenges in MA chemistry, especially when aiming to combine an efficient biotechnological production of MA with its valorization.
![[50] Synthesis-structure-activity relations in Fe-CHA for C-H activation: control of Al-distribution by interzeolite conversion](https://dusselier-lab.org/uploads/media/cache/default/uploads/ee488d2d3d457a829c7759b95b1d8bcd.jpeg)
[50] Synthesis-structure-activity relations in Fe-CHA for C-H activation: control of Al-distribution by interzeolite conversion 12-01-2020
The search for structurally relevant Al-arrangements in zeolites is an important endeavor for single site catalysis. Little is known about the mechanisms and zeolite dynamics during synthesis that are responsible for creating those Al-ensembles. Here, new synthetic strategies for creating Al-hosts in small-pore zeolites suitable for divalent cation catalysis are uncovered, leading to a mechanistic proposal for Al-organization during crystallization. As such, unique synthesis-structure-activity relations are demonstrated for the partial oxidation of methane on Fe-exchanged CHA-zeolites. With modified interzeolite conversions, the divalent cation capacity of the resulting high Si SSZ-13 zeolites (Si/Al ~ 35) can be reproducibly controlled in a range between 0.04 and 0.34 Co2+/Al. This capacity is a proxy for the distribution of framework aluminum in pairs and correlates with the methanol production per Al when these zeolites host the α-FeII redox active site. The uncovered IZC synthesis-structure relations paint an Al-distribution hypothesis, where incongruent dissolution of the starting USY zeolite and fast synthesis kinetics with atypical growth modes allow assembling specific Al-arrangements, resulting in a high divalent cation capacity. Prolonged synthesis times and high temperatures overcome the energetic barriers for T-atom reshuffling favoring Al-isolation. These mechanisms and the relations uncovered in this work will guide the search for relevant Al-ensembles in a range of zeolite catalysts where controlling the environment for a single active site is crucial.
2019
![[49] Substrate-specificity of Candida rugosa lipase and its industrial application](https://dusselier-lab.org/uploads/media/cache/default/uploads/31917ec46c47695c94e27744cf5bd47a.jpeg)
[49] Substrate-specificity of Candida rugosa lipase and its industrial application 11-09-2019
Candida rugosa lipase (CRL) is a very versatile and widely used lipase in the aroma and flavor industry, the fat and oil industry, the pharmaceutical industry and is also applicable as a biosensor. The present paper discusses recent developments on the molecular level of CRL, the biocatalytic reactivity of several Lip isoenzymes, diverse immobilization strategies of the enzyme and its broad (substrate) specificities in different fields. Furthermore, an overview is given of future applications with CRL such as the enantioselective hydrolysis of racemic lactate esters produced out of sugars and the production of biodiesel out of vegetable oil. These processes have been tried on lab scale but still need further optimization for industrialization.
![[48] Sustainable Chemistry Considerations for the Encapsulation of Volatile Compounds in Laundry-type Applications](https://dusselier-lab.org/uploads/media/cache/default/uploads/4f6ba5a2f085b9851009a4c496e946dd.png)
[48] Sustainable Chemistry Considerations for the Encapsulation of Volatile Compounds in Laundry-type Applications 05-04-2019
Microencapsulation of volatile compounds in fabric care products has brought extra value in a variety of laundry-type applications, allowing clothes to release pleasant scents for weeks after their last wash with minimal amounts of fragrance used. Melamine-formaldehyde is the industry standard in this regard, but polyacrylate and polyurea are also used as micro-capsule wall material in commercial laundry-type applications. Harsh storage conditions and demanding release characteristics have limited the number of viable shell wall materials and chemistries for these kinds of applications. This renders nano- and microencapsulation of volatile compounds for laundry-type applications one of the most challenging areas in the encapsulation field. The largest drawback of the current technology is the limited biodegradability of the produced microcapsules, e.g. when leaking via waste water. This review summarizes the search towards viable, high-performant and sustainable alternatives for the current technology. First, various techniques to encapsulate volatile compounds in this context are overviewed. Recent relevant encapsulation reports using natural and synthetic shell walls are discussed, while controlled release data are included where possible. Finally, a perspective containing insights toward sustainability in the engineering of alternative capsule chemistries is offered.
![[47] Aerosol route to TiO2–SiO2 catalysts with tailored pore architecture and high epoxidation activity](https://dusselier-lab.org/uploads/media/cache/default/uploads/18d5986d6b8889d9822f692329f826cf.jpeg)
[47] Aerosol route to TiO2–SiO2 catalysts with tailored pore architecture and high epoxidation activity 25-01-2019
Herein, we present the aerosol-assisted sol-gel preparation of hierarchically porous TiO2–SiO2 catalysts having a sphere-like shell morphology and a high Ti dispersion. In order to control the porosity at the micro-, meso- and macro- levels, we use the evaporation-induced self assembly (EISA) of a surfactant, possibly combined with polymer beads as hard templates. These catalysts are tested for the epoxidation of cyclohexene with cumene hydroperoxide as oxidant, and their performance is compared to the reference TS-1 zeolite. The high catalytic performance observed with the catalysts prepared by aerosol stems from their high specific surface area, but also from the short diffusion path length generated by the meso-/macro-pore architecture which provide entryways for bulky reactants and products. Besides, these materials can incorporate a higher Ti loading than TS-1 zeolite, while ensuring a good control over the Ti speciation. Thus, the unique features of the aerosol pro-cess – which is also known to be scalable – allow us to prepare catalytic materials with high epoxidation activity, also for bulky olefins.
![[46] MOF-derived metal oxide clusters in porous aluminosilicates: a new catalyst design for the synthesis of bioactive aza-heterocycles](https://dusselier-lab.org/uploads/media/cache/default/uploads/739ea69f405659abc1c3f4ad55e6c6bd.jpeg)
[46] MOF-derived metal oxide clusters in porous aluminosilicates: a new catalyst design for the synthesis of bioactive aza-heterocycles 03-01-2019
Simple solid-state mixing and calcination of catalytic amounts of metal-carboxylate frameworks in the presence of aluminosilicates, allows for the generation of novel active and robust supported metal oxide nanoparticles that catalyze C-C and C-N bond formations. The proposed Cu and Zn containing aluminosilicates outperform state-of-the-art solid catalysts for the synthesis of various aza-heterocycles under mild and heterogeneous conditions, exhibiting the highest TOFs ever reported for cost-efficient and reusable CuO and ZnO.
![[45] Bio‐Acrylates Production: Recent Catalytic Advances and Perspectives of the Use of Lactic Acid and their Derivatives](https://dusselier-lab.org/uploads/media/cache/default/uploads/ebc3979c5c3c3873211c985051554b97.jpeg)
[45] Bio‐Acrylates Production: Recent Catalytic Advances and Perspectives of the Use of Lactic Acid and their Derivatives 01-01-2019
The production of drop‐in chemicals from bio‐based renewable sources is gaining a lot of momentum due to proven negative impact of fossil‐based economy on environment and society. In this review, various bio‐derived platform molecules are assessed as renewable alternatives to fossil resources for the catalytic production of acrylates. Acrylic acid and its esters are key building blocks of a large number of high‐value oligomers and polymers in the current industry. In spite of the encouraging successes reported on gram or lab‐scale, real implementation of bio‐based examples remain scarce mainly due to the current high cost and limited availability of the bio‐based substrates. As lactic acid and their derivatives are one of the most promising feedstocks for bio‐acrylate production, they are the main focus of this review.
2018
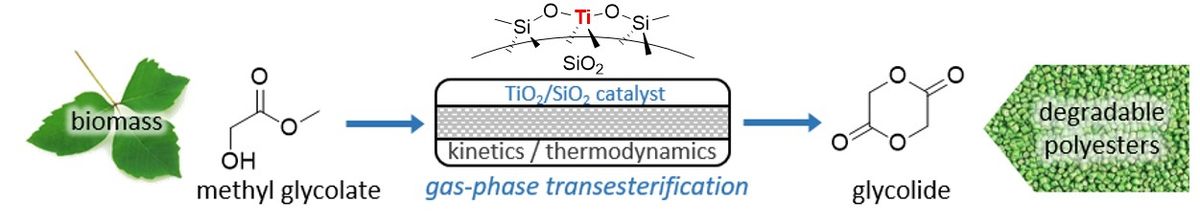
Catalytic Gas‐Phase Cyclization of Glycolate Esters: a Novel Route Toward Glycolide‐Based Bioplastics 29-10-2018
A catalytic process to produce glycolide, the cyclic dimer of glycolic acid (GA), is proposed. Glycolide is the key building block of the biodegradable plastic polyglycolic acid. Instead of the current industrial two‐step route, which involves the polycondensation of GA and a subsequent backbiting reaction, a new route based on the gas‐phase transesterification of methyl glycolate (MGA) over a fixed catalyst bed is presented. With specific supported TiO2 catalysts, a high glycolide selectivity of 75‐78% can be achieved at the thermodynamically‐limited equilibrium conversion of MGA (54% at 300°C, 5.6vol% MGA). The absence of solvent and the continuous nature of the process should allow for easy product separation and recycling of unconverted esters, while the few side‐products, i.e. linear alkyl glycolate dimers and trimers seem recoverable via methanolysis. The reaction is compared to the cyclization of other α‐hydroxy esters, such as methyl lactate to lactide, over the same catalysts, in terms of kinetics and thermodynamics. The absence of a methyl substitution on the α‐carbon seems to lead to faster cyclization kinetics of MGA when compared to methyl lactate or the double‐substituted methyl‐2‐hydroxy‐isobutyrate. Contrarily, glycolide production is less favored thermodynamically compared to lactide. The absence of glycolide decomposition at temperatures up to 300°C however allows to increase equilibrium conversion by taking the endergonic reaction to higher temperatures.

Direct upstream integration of biogasoline production into current light straight run naphtha petrorefinery processes. 27-09-2018
There is an urgent need to address environmental problems caused by our transportation systems, which include the reduction of associated CO2 emissions. In the short term, renewable drop-in fuels are ideal, as they allow a direct integration into the existing infrastructure. However, preferably they would perform better than current alternatives (for example, bioethanol) and be synthesized in a more efficient way. Here we demonstrate the production of biogasoline with a direct upstream integration into processes in existing petrorefinery facilities that targets the 10% bio-based carbon in accordance with the current European Union directives (for 2020) for biofuels. To achieve this goal, we show the valorization of (hemi)cellulose pulp into light naphtha using a two-phase (H2O:organic) catalytic slurry process. A C5–C6alkane stream, enriched with bio-derived carbon and compatible with further downstream petrorefinery operations for (bio)gasoline production, is automatically obtained by utilizing fossil light straight run naphtha as the organic phase. The ease of integration pleads for a joint petro/bio effort to gradually produce bio-enriched gasolines, wherein the chemical compounds of the bio-derived fraction are indistinguishable from those in current high-quality gasoline compositions.

Spectroscopic Identification of the α-Fe / α-O Active Site in Fe-CHA Zeolite for the Low-Temperature Activation of the Methane C-H bond 01-09-2018
The formation of single-site α-Fe in the CHA zeolite topology is demonstrated. The site is shown to be active in oxygen atom abstraction from N2O to form a highly reactive α-O, capable of methane activation at room temperature to form methanol. The methanol product can subsequently be desorbed by on-line steaming at 200°C. For the intermediate steps of the reaction cycle, the evolution of the Fe active site is monitored by UV-Vis-NIR and Mössbauer spectroscopy. A B3LYP-DFT model of the α-Fe site in CHA is constructed and the ligand field transitions are calculated by CASPT2. The model is experimentally substantiated by the preferential formation of α-Fe over other Fe species, the requirement of paired framework aluminium and a MeOH/Fe ratio indicating a mononuclear active site. The simple CHA topology is shown to mitigate the heterogeneity of iron speciation found on other Fe-zeolites, with Fe2O3 being the only identifiable phase other than α-Fe formed in Fe-CHA. The α-Fe site is formed in the d6r composite building unit, which occurs frequently across synthetic and natural zeolites. Finally, through a comparison between α-Fe in Fe-CHA and Fe-*BEA, the topology’s 6MR geometry is found to influence the structure, the ligand field, and consequently the spectroscopy of the α-Fe site in a predictable manner. Variations in zeolite topology can thus be used to rationally tune the active site properties.
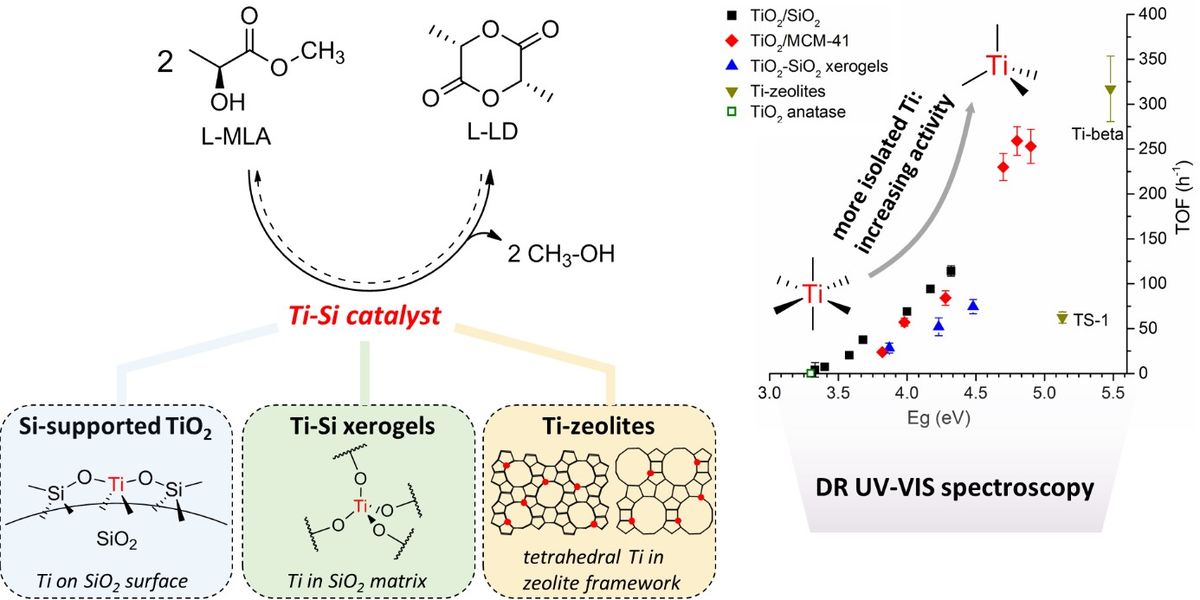
Titania-Silica Catalysts For Lactide Production From Renewable Alkyl Lactates: Structure-Activity Relations 24-07-2018
Different Ti-Si catalysts, viz. TiO2 supported on amorphous SiO2 or Si-MCM-41, TiO2-SiO2 xerogels and Ti-zeolites (TS-1 and Ti-beta) were compared in terms of activity and selectivity for the direct conversion of methyl lactate to lactide in the gas phase. Except for Ti-beta, all catalysts exhibit a high lactide selectivity of 88-92% at conversions below 50 %. From DR UV-VIS spectroscopy, it is evidenced that the catalytic activity of tetrahedral TiO4 sites is higher than of polymerized TiO5 or octahedral TiO6 counterparts, irrespective of the catalyst structure, an analysis supported by ToF-SIMS measurements. A kinetic analysis shows that the catalytic activity is proportional to the number of vacant sites on the catalyst surface. Thus, the activity increase observed for tetrahedral TiO4 sites may be attributed to an increased number of vacant sites (e.g. two for TiO4, zero for TiO6). Lactide productivity thus highly benefits from an increased dispersion of Ti-sites on the catalyst surface, and could be increased by a factor of 2.5 (up to 10 gLD gcat-1 h-1) when TiO2 is dispersed on a Si-MCM-41 support, with higher surface areas than amorphous SiO2 gels.
One-step synthesis of stereo-pure l,l lactide from l-lactic acid 13-06-2018

Small-Pore Zeolites: Synthesis and Catalysis 13-05-2018
In the past decade or so, small-pore zeolites have received greater attention than large- and medium-pore molecular sieves that have historically dominated the literature. This is primarily due to the commercialization of two major catalytic processes, NOx exhaust removal and methanol conversion to light olefins, that take advantage of the properties of these materials with smaller apertures. Small-pore zeolites possess pores that are constructed of eight tetrahedral atoms (Si4+and Al3+), each time linked by a shared oxygen These eight-member ring pores (8MR) provide small molecules access to the intracrystalline void space, e.g., to NOx during car exhaust cleaning (NOx removal) or to methanol en route to its conversion into light olefins, while restricting larger molecule entrance and departure that is critical to overall catalyst performance. In total, there are forty-four structurally different small-pore zeolites. Forty-one of these zeolites can be synthesized, and the first synthetic zeolite (KFI, 1948) was in fact a small-pore material. Although the field of 8MR zeolite chemistry has expanded in many directions, the progress in synthesis is framework-specific, leaving insights and generalizations difficult to realize. This review first focuses on the relevant synthesis details of all 8MR zeolites and provides some generalized findings and related insights. Next, catalytic applications where 8MR zeolites either have been commercialized or have dominated investigations are presented, with the aim of providing structure–activity relationships. The review ends with a summary that discusses (i) both synthetic and catalytic progress, (ii) a list of opportunities in the 8MR zeolite field, and (iii) a brief future outlook.
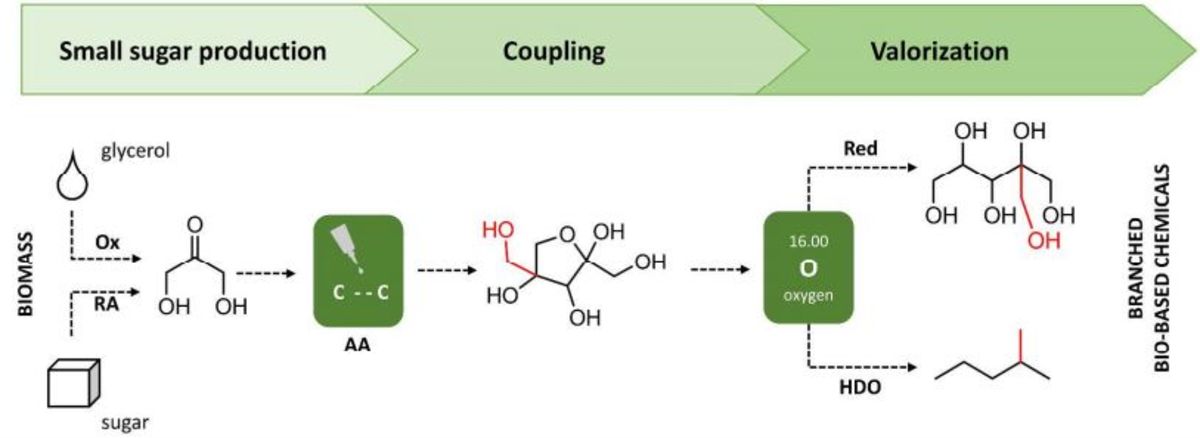
Branching-first: synthesizing C-C skeletal branched bio-based chemicals from sugars 26-04-2018
A novel strategy to bio-based chemicals with a branched carbon skeleton is introduced. Hereto, small sugars, such as 1,3-dihydroxyacetone, are coupled catalytically to obtain branched C6 sugars, such as dendroketose, in high yield at mild conditions. By bringing this branching step up front, at the level of the sugar feedstock (branching-first), new opportunities for the synthesis of useful chemicals arise. Here, we show that the branched sugar can be efficiently valorized into (i) new branched polyols and (ii) short branched alkanes. The first route preserves most of the original sugar functionality by hydrogenation with Ru/C, and renders access to branched polyols with three primary alcohol groups. These molecules are potentially interesting as plasticizers, crosslinkers or detergent precursors. The second valorization route demonstrates a facile hydrodeoxygenation of the branched sugars towards their corresponding branched alkanes (e.g. 2-methylpentane). The highest alkanes yields (> 65 mol% C) are obtained with a Rh/C redox metal catalyst in a biphasic catalytic system, following a HDO mechanism. In the short term, commercial integration of these mono-branched alkanes, in contrast to branched polyols, is expected to be straightforward, because of their drop-in character and well-known valuable octane booster role when present in gasoline. Accordingly, the branching-first concept is also demonstrated with other small sugars (e.g. tetroses) enabling the production of branched C6-C8 sugars, and thus also branched C5-C8 alkanes after HDO.
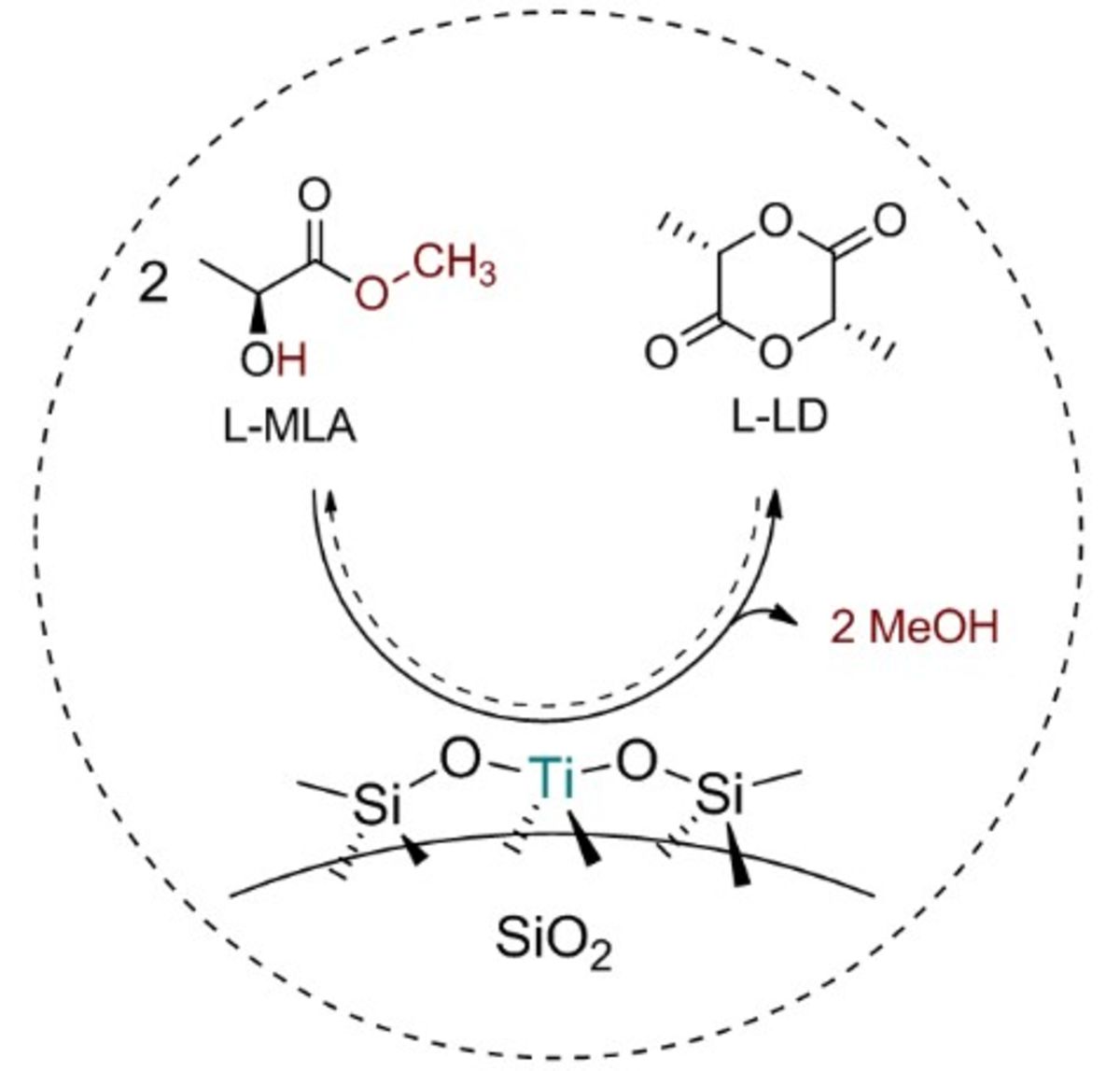
Catalytic gas-phase production of lactide from renewable alkyl lactates 20-01-2018
A new route to lactide, key building block of the bioplastic polylactic acid, is proposed via a continuous catalytic gas-phase transesterification of renewable alkyl lactates in a scalable fixed-bed setup. Supported TiO2/SiO2 catalysts are highly selective to lactide, with only minimal lactide racemization. The solvent-free process allows for easy product separation and recycling of unconverted alkyl lactates and recyclable lactyl intermediates. The catalytic activity of TiO2/SiO2 catalysts was strongly correlated to their optical properties by DR UV-VIS spectroscopy. Catalysts with high band gap energy of the supported TiO2 phase, indicative of a high surface spreading of isolated Ti centers, show the highest turnover frequency per Ti site.

Synthetic and Catalytic Potential of Amorphous Mesoporous Aluminosilicates Prepared by Postsynthetic Aluminations of Silica in Aqueous Media 01-01-2018
Amorphous aluminosilicate catalysts have been used industrially on a large scale for almost a century. However, the influence of the pH on the alumination of silica in aqueous solutions has remained largely unclear. Herein, room temperature aluminations of different mesoporous amorphous silicas (fumed silica, dried silica gel, SBA‐15, MCM‐41, and COK‐12) with aqueous solutions of various pH (3–13) are explored. The aqueous solutions are prepared using different aluminum sources (Al(NO3)3 or NaAlO2) and alkaline additives (NaOH or NH4OH). The decoupling of pH and Al source using alkaline additives results in a vast experimental potential to prepare unique aluminosilicates, whereby an important role is played by the pH development during the treatment. The bulk and surface composition, acidity, aluminum coordination, morphology, hydrothermal stability, and porosity of the obtained materials are characterized. Optimal samples possess large surface areas and superior acidities (up to 50 % higher) and outstanding stabilities compared to aluminosilicates prepared with state‐of‐the‐art methods. The obtained materials are evaluated in a series of acid‐catalyzed model reactions involving substrates of various chemical reactivity and size, enabling insight in the catalytic functionality of the introduced Brønsted and Lewis sites. The potential of the obtained materials is emphasized by the similar or superior acidity and catalytic performance compared to several benchmark industrial silica–alumina‐based catalysts.
2017
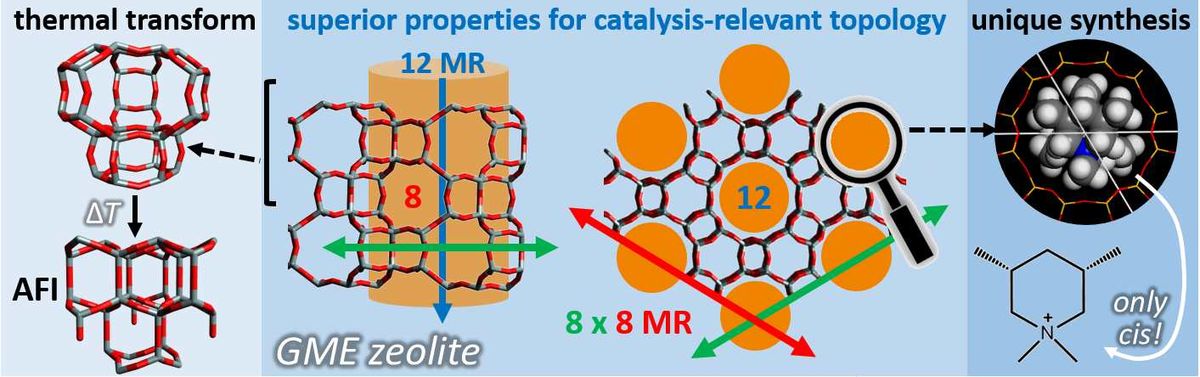
CIT-9: A Fault-Free Gmelinite Zeolite 11-10-2017
This can be considered the first paper published since the start of the autonomous research group.
It was included in the online issue on October 11, 2017
synthetic, fault-free gmelinite (GME) zeolite is prepared using a specific organic structure-directing agent (OSDA), cis-3,5-dimethylpiperidinium. The cis-isomers align in the main 12-membered ring (MR) channel of GME. Trans-isomer OSDA leads to the small-pore zeolite SSZ-39 with the OSDA in its cages. Data from N2-physisorption and rotation electron diffraction provide evidence for the openness of the 12 MR channel in the GME 12×8×8 pore architecture and the absence of stacking faults, respectively. CIT-9 is hydrothermally stable when K+-exchanged, while in the absence of exchange, the material transforms into an aluminous AFI-zeolite. The process of this phase-change was followed by in situ variable temperature powder X-ray diffraction. CIT-9 has the highest Si/Al ratio reported for GME, and along with its good porosity, opens the possibility of using GME in a variety of applications including catalysis.

Heterogeneous catalysis for bio-based polyester monomers from cellulosic biomass: advances, challenges and prospects 07-09-2017
It is a 21st century challenge to develop a more sustainable chemical industry where fossil-based resources are, where possible, preferentially replaced by renewable alternatives. Bio-based polymers, in particular those derived from cellulose or other carbohydrates, are often considered benign alternatives for petrochemical plastics. The majority of bioplastic precursors are currently derived from fermentation or biotechnology. Chemocatalytic routes to both similar and new polymer building blocks are emerging in an effort to mitigate challenges related to carbohydrate fermentation, such as waste generation and costly product purification. This review critically surveys recent developments in applying heterogeneous catalysis for the production of bio-based polyester monomers from cellulose or cellulose-derived carbohydrates. Highlighted target molecules include various α-hydroxy acids or esters (e.g. lactic and glycolic acid, lactide and methyl vinyl glycolate), furandicarboxylic acid, ethylene glycol and isosorbide. The production of lactic acid from glycerol will exceptionally be included as well, as an oversupply of glycerol might contribute to non-negligible amounts of lactic acid in the future. Where possible, remaining challenges and future prospects are highlighted.
Lewis acid catalysis on single site Sn centers incorporated into silica hosts 29-05-2017
Tetrahedral Sn built into microporous silica frameworks such as zeolites and structured mesoporous silica can be used as heterogeneous Lewis acid catalysts. These materials have recently attracted much attention, as they show remarkable activity and selectivity in a wide range of reactions. A prominent example is the conversion of carbohydrates into platform and commodity chemicals such as lactic acid or alkyl lactates, where the activity and selectivity of Sn-based materials remains unsurpassed compared to Sn-free catalysts. Some of the materials show water-tolerant behavior and can therefore also be used in aqueous systems. In this work, a literature overview regarding synthesis of Sn-containing silica materials is given, as well as a synopsis of the characterization tools which can be used to unravel the structure of the catalytic active site. The application of such Sn-containing materials for diverse catalytic reactions is reviewed, with special emphasis on the effects of the catalyst characteristics on the catalytic activity and stability.
Structural and Kinetic Changes to Small-Pore Cu-Zeolites After Hydrothermal Aging Treatments and Selective Catalytic Reduction of NOx with Ammonia 01-01-2017
Three small-pore, eight-membered ring (8-MR) zeolites of different cage-based topology (CHA, AEI, RTH), in their proton- and copper-exchanged forms, were first exposed to high temperature hydrothermal aging treatments (1073 K, 16 h, 10% (v/v) H2O) and then to reaction conditions for low temperature (473 K) standard selective catalytic reduction (SCR) of NOxwith ammonia, in order to study the effect of zeolite topology on the structural and kinetic changes that occur to Cu-zeolites used in NOxabatement. UV-visible spectra were collected to monitor changes to Cu structure and showed that band intensities for isolated, hydrated Cu2+cations (∼12 500 cm−1) remain constant after hydrothermal aging, but decrease in intensity upon subsequent exposure to low temperature SCR reaction conditions. Standard SCR rates (per Cu, 473 K), activation energies, and reaction orders are similar between Cu-AEI and Cu-CHA zeolites before and after hydrothermal aging, although rates are lower after hydrothermal aging as expected from the decreases in intensity of UV-visible bands for Cu2+ active sites. For Cu-RTH, rates are lower (by 2–3×) and apparent activation energies are lower (by ∼2×) than for Cu-AEI or Cu-CHA. These findings suggest that the RTH framework imposes internal transport restrictions, effectively functioning as a one-dimensional framework during SCR catalysis. Hydrothermal aging of Cu-RTH results in complete deactivation and undetectable SCR rates, despite X-ray diffraction patterns and Ar micropore volumes (87 K) that remain unchanged after hydrothermal aging treatments and subsequent SCR exposure. These findings highlight some of the differences in low temperature SCR behavior among small-pore Cu-zeolites of different topology, and the beneficial properties conferred by double six-membered ring (D6R) composite building units. They demonstrate that deleterious structural changes to Cu sites occur after exposure to hydrothermal aging conditions and SCR reactants at low temperatures, likely reflecting the formation of inactive copper-aluminate domains. Therefore, the viability of Cu-zeolites for practical low temperature NOx SCR catalysis cannot be inferred solely from assessments of framework structural integrity after hydrothermal aging treatments, but also require Cu active site and kinetic characterization after hydrothermally aged zeolites are exposed to low temperature SCR reaction conditions.
2016
Compositional and structural feedstock requirements of a liquid phase cellulose-to-naphtha process in a carbon- and hydrogen-neutral biorefinery context 27-07-2016
Processing raw (ligno)cellulosic feedstock into renewable light naphtha alkanes could lead to a gradual replacement of fossil feedstock for the production of chemicals, materials and fuels. The production of drop-in alkanes is a preferable short term strategy because of its practical implementation and integration in existing infrastructure and processes. A handful of promising cellulose-to-alkane biorefinery initiatives were recently reported, both processing in gas and liquid phase. This contribution presents a detailed study of the two-liquid phase hydrodeoxygenation of cellulose to n-hexane under relatively mild circumstances, proceeding through the recently communicated HMF route, in presence of a soluble acid and Ru/C metal catalyst. Two main points were addressed here: (i) the importance (or not) of the lignocellulose pretreatment and purification to the alkane yield, and (ii) the renewability of the consumed hydrogen in the process. A systematic study of the effect of cellulose purity, crystallinity, degree of polymerization and particle size (surface area) on the light naphtha yield was performed to tackle the first part. As fibrous cellulose with large particles was the most favourable feedstock with regard to alkane yield and as the presence of hemicellulose and lignin impurities had no effect on the cellulose-to-naphtha conversion, costly mechanical and purification steps are redundant to the process, in contrast to their notable importance in other cellulose valorisation processes (e.g. to glucose, sorbitol, isosorbide and acids). The second point regarding sustainable hydrogen supply is discussed in detail by calculating hydrogen and carbon mass and energy balances of the chemical conversions, assuming selected scenarios among others to recuperate the hydrogen by steam-reforming of waste streams (like gaseous C<6 hydrocarbons and aqueous polyol fractions) and (partial) aromatization of the C6 fraction into benzene. The study shows potential to integrate the liquid phase cellulose-to-naptha (LPCtoN) technology into a self-sufficient biorefinery, in which the chemical processes may run without consumption of external (non-renewable) hydrogen, carbon and energy, except for solar light.
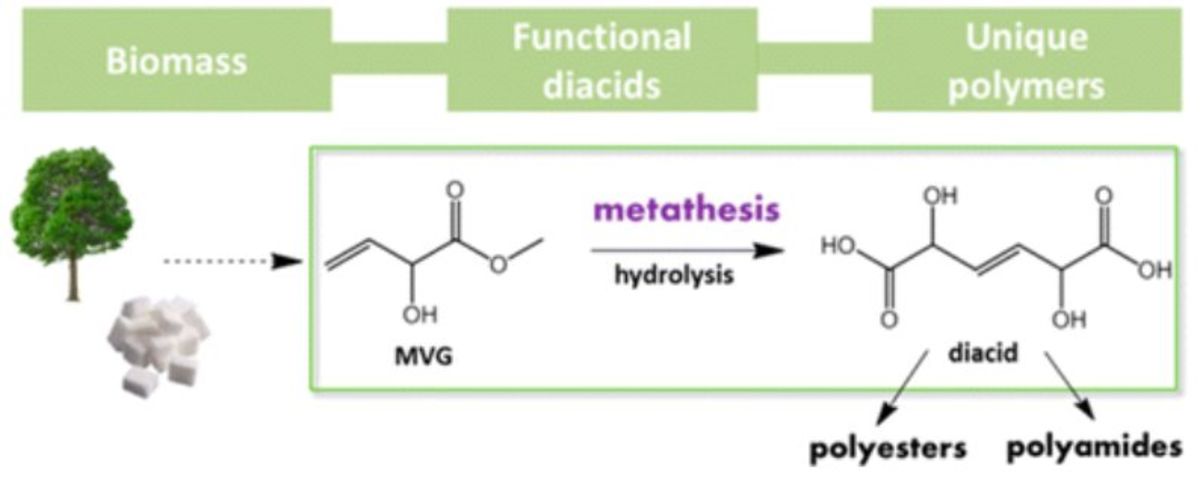
Synthesis of Novel Renewable Polyesters and Polyamides with Olefin Metathesis 31-05-2016
Unsaturated and hydroxyl-functionalized C6-dicarboxylic acids were successfully synthesized via olefin metathesis from methyl vinyl glycolate (MVG), a renewable α-hydroxy C4-ester product from Lewis-acid carbohydrate conversion. Addition of a second-generation Hoveyda–Grubbs catalyst to neat MVG leads to a near quantitative yield of dimethyl-2,5-dihydroxy-3-hexenedioate (DMDHHD). Additional hydrolysis and hydrogenation steps form interesting polymer building blocks like 2,5-dihydroxy-3-hexenedioic acid (DHHDA) and 2,5-dihydroxyadipic acid (DHAA). Their use in polyester and polyamide synthesis is demonstrated after determination of their physical and spectroscopic characteristics. Copolymerization of DHHDA with l-lactic acid for instance produces a cross-linked poly(l-lactic acid-co-DHHDA) polyester. Proof of cross-links is ascertained by NMR and FTIR. Substantial impact on the melting, thermal, and polar properties of PLA are observed already at low amounts of DHHDA (0.1 mol %) in accord with the presence of cross-links in the polymer. Biobased polyamides were also synthesized by equimolar reaction of DHHDA with hexamethylenediamine, producing a renewable polyamide analogue of the petroleum-based nylon-6,6. Interestingly, the as-synthesized polyamide (α-bishydroxylated unsaturated polyamide, HUPA) possesses similar thermal stability as nylon-6,6 but shows different chemical properties as a result of the double bond and α-hydroxy functionality.
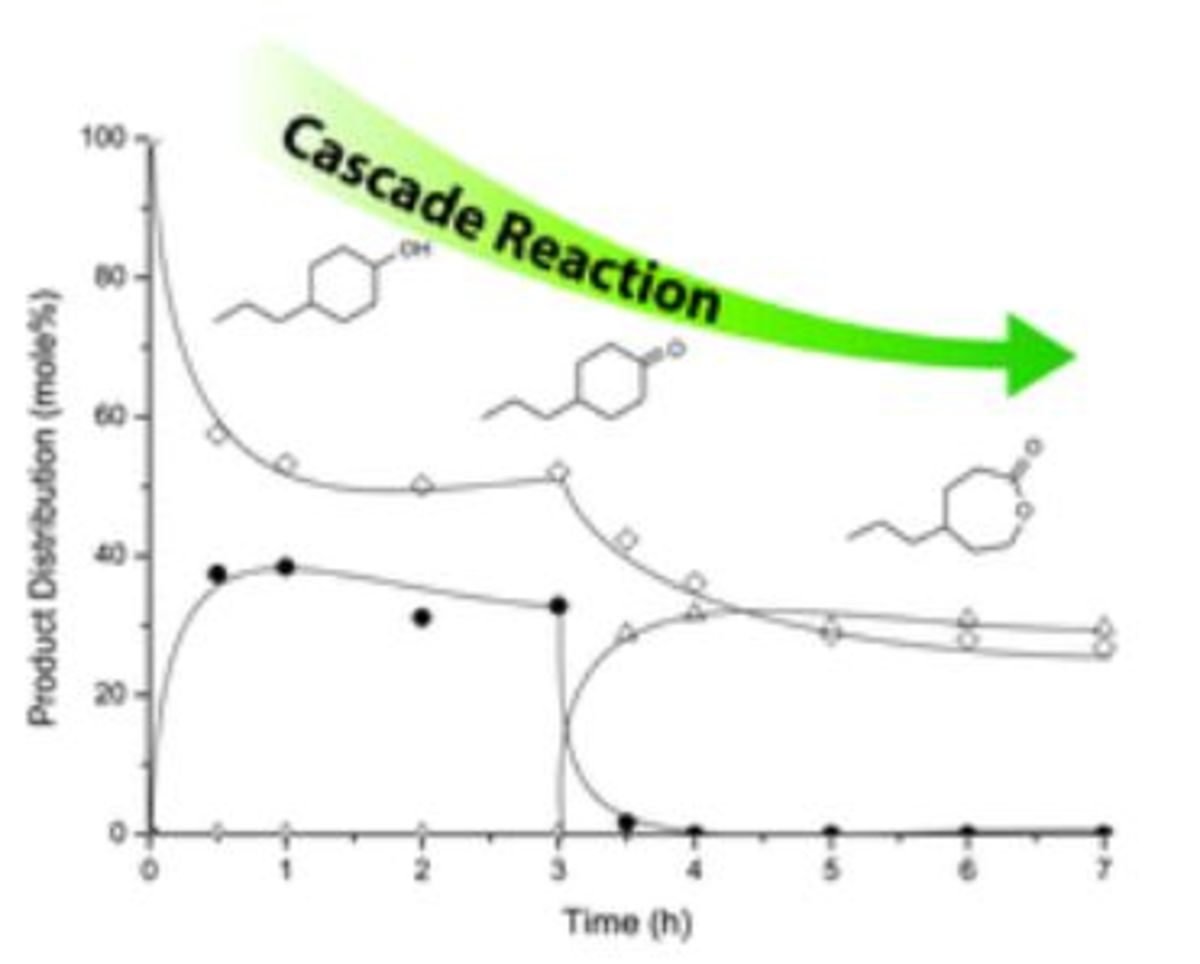
Snβ-zeolite catalyzed oxido-reduction cascade chemistry with biomass-derived molecules 18-04-2016
High activity of post-synthetically synthesized Sn-beta, producing novel caprolactone polymer building blocks, is demonstrated in Meerwein–Ponndorf–Verley (MPV), Oppenauer (OPO), Baeyer–Villiger (BV) and cascade reactions thereof with biomass-derived molecules.
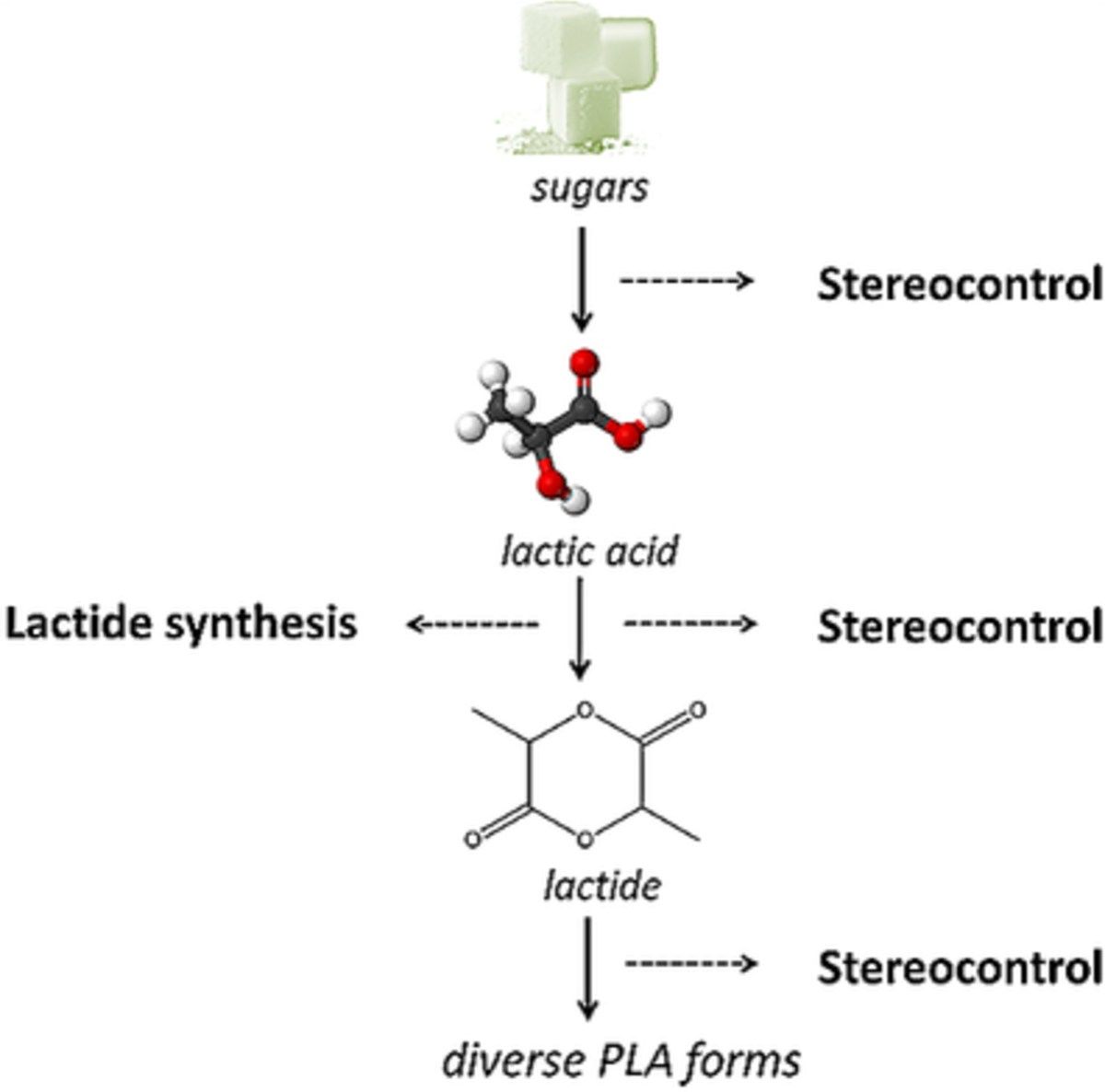
Lactide Synthesis and Chirality Control for Polylactic acid Production 01-03-2016
Polylactic acid (PLA) is a very promising biodegradable, renewable, and biocompatible polymer. Aside from its production, its application field is also increasing, with use not only in commodity applications but also as durables and in biomedicine. In the current PLA production scheme, the most expensive part is not the polymerization itself but obtaining the building blocks lactic acid (LA) and lactide, the actual cyclic monomer for polymerization. Although the synthesis of LA and the polymerization have been studied systematically, reports of lactide synthesis are scarce. Most lactide synthesis methods are described in patent literature, and current energy-intensive, aselective industrial processes are based on archaic scientific literature. This Review, therefore, highlights new methods with a technical comparison and description of the different approaches. Water-removal methodologies are compared, as this is a crucial factor in PLA production. Apart from the synthesis of lactide, this Review also emphasizes the use of chemically produced racemic lactic acid (esters) as a starting point in the PLA production scheme. Stereochemically tailored PLA can be produced according to such a strategy, giving access to various polymer properties.
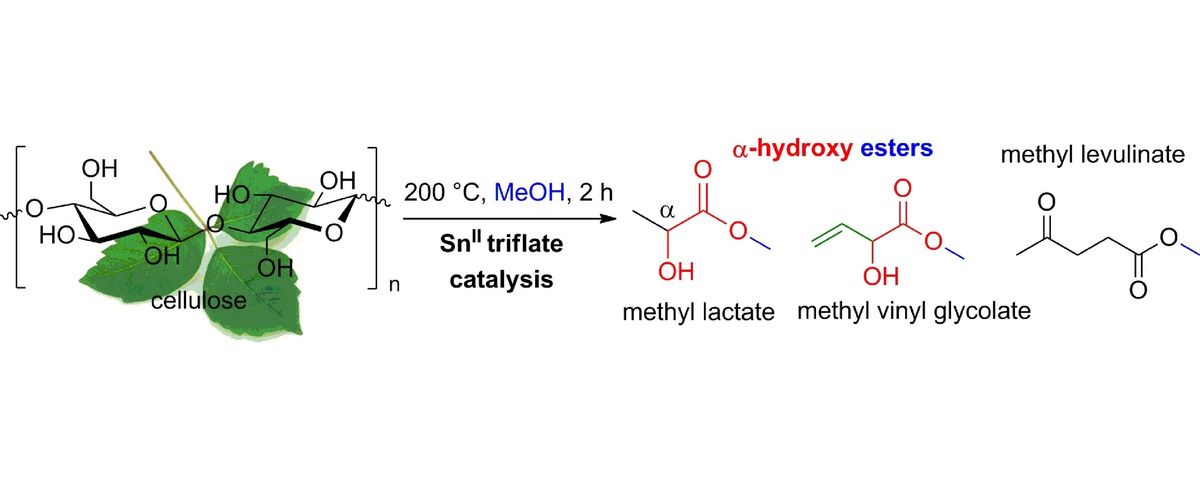
Tin triflate-catalyzed conversion of cellulose to valuable (α-hydroxy-) esters 15-02-2016
The direct conversion of cellulose with metal-triflate catalysts in methanol is investigated. SnII-triflate remarkably catalyzes the formation of a mixture of useful α-hydroxy esters such as methyl lactate, methyl vinyl glycolate and methyl-4-methoxy-2-hydroxybutanoate, on top of methyl levulinate. Compared to other metal triflates or Sn salts, the catalytic features of SnII-triflate are distinct and linked to the interplay between its Brønsted and Lewis acidic component. A total ester yield (carbon-based, mol%) in the 60%-range could be obtained from cellulose after 2 h at 200 °C for cellulose loadings up to 20 g L−1 with 4.8 mM of catalyst. The cascade reaction network, confirmed by feeding intermediates, highlights the importance of a fast retro-aldol of the hexose intermediates – opposed to their dehydration – when α-hydroxy esters are targeted. By manipulating the triflate-to-Sn ratio, nearly 40% of α-hydroxy esters can be produced in a one-pot approach. Such mixtures could help fuel the demand for functional biodegradable polyesters.
Potential and challenges of zeolite chemistry in the catalytic conversion of biomass 01-01-2016
Increasing demand for sustainable chemicals and fuels has pushed academia and industry to search for alternative feedstocks replacing crude oil in traditional refineries. As a result, an immense academic attention has focused on the valorisation of biomass (components) and derived intermediates to generate valuable platform chemicals and fuels. Zeolite catalysis plays a distinct role in many of these biomass conversion routes. This contribution emphasizes the progress and potential in zeolite catalysed biomass conversions and relates these to concepts established in existing petrochemical processes. The application of zeolites, equipped with a variety of active sites, in Brønsted acid, Lewis acid, or multifunctional catalysed reactions is discussed and generalised to provide a comprehensive overview. In addition, the feedstock shift from crude oil to biomass involves new challenges in developing fields, like mesoporosity and pore interconnectivity of zeolites and stability of zeolites in liquid phase. Finally, the future challenges and perspectives of zeolites in the processing of biomass conversion are discussed.

An Inner-/Outer-Sphere Stabilized Sn Active Site in β-Zeolite: Spectroscopic Evidence and Kinetic Consequences. 01-01-2016
A highly active Sn site with Lewis acid properties is identified in post-synthetically synthesized Sn/DeAlβ catalyst, prepared by liquid-phase Sn grafting of a dealuminated β-zeolite. Though apparently similar Sn active-site structures have been reported for the post-synthetic and the conventional hydrothermal Snβ, detailed study of the electronic structure and redox behavior of Sn with EXAFS, XANES, DR UV–vis, and TPR clearly reveals dissimilarities in geometry and electronic properties. A model of the active Sn site is proposed using a contemporary interpretation of inner-/outer-sphere coordination, assuming inner-sphere coordination of SnIV with three framework SiO– and one outer-sphere coordination by a distant charge-balancing SiO–, resulting in a separated Lewis acid–base pair. Stabilization of this geometry by a nearby water molecule is proposed. In comparison with active Sn sites in a hydrothermally synthesized Snβ, those in the grafted dealuminated material are sterically less demanding for substrate approach, while the low inner-sphere coordination of Sn leads to a stronger Lewis acidity. Proximate silanols in the active-site pocket, identified by FTIR, 29Si MAS NMR, 1H–29Si CP MAS NMR, DR NIR, and TGA, may impact local reagent concentration and transition states stabilization by hydrogen bonding. The structural dissimilarity of the active Sn site leads to a different kinetic behavior. Kinetic experiments using two Lewis-acid-catalyzed reactions, Baeyer–Villiger and Meerwein–Ponndorf–Verley, show differences that are reaction-type dependent and have different entropic (like sterical demand and hydrogen bonding) and enthalpic contributions (Lewis acid strength). The active-site model, containing both inner- and outer-sphere ligands with the zeolite framework, may be considered as a general model for other grafted Lewis acid single sites.
2015
Methanol-to-Olefins Catalysis with Hydrothermally Treated Zeolite SSZ-39. 17-09-2015
Zeolite SSZ-39 is evaluated for catalyzing the methanol-to-olefins (MTO) reaction. By steaming NH4–SSZ-39, Al can be removed from framework positions, resulting in an increase in framework-Si/AlT and thus a lowered active acid site density. The Si/AlT ratios can be controlled by the steaming temperatures. SSZ-39 steamed at 750 °C, with preserved pore volume and morphology, is an excellent MTO catalyst, as high, stable olefin selectivities, long time-on-stream activity, and low alkane production are observed. Moreover, interesting propylene/ethylene/butylene ratios of 2.8/1/1.1 are obtained, likely related to the shape of the AEI cage. By Cu2+-exchanging SSZ-39, evidence is provided to show that AlT sites in close proximity (high AlT density) produce the unwanted effects (higher alkane-make and carbonaceous deposits) in nonsteamed materials during MTO.
Confinement Effects in Lewis Acid-Catalyzed Sugar Conversion: Steering toward Functional Polyester Building Blocks 24-08-2015
We report the use of solid Lewis acid catalysts for the conversion of tetrose sugars to four-carbon α-hydroxy acid esters (C4-AHA), which are useful as functional polyester building blocks. Sn-β was by far the most active and selective catalyst, yielding up to 80% methyl vinyl glycolate (MVG), methyl-4-methoxy-2-hydroxybutanoate (MMHB), and α-hydroxy-γ-butyrolactone (HBL) combined at 95% conversion. A very high turnover frequency (TOF) of 330 molC4-AHA molSn h–1 was attained using Sn-β, a more than 6-fold increase compared with homogeneous SnCl4·5H2O. It is shown that, using different Sn-based catalysts with various pore sizes, the product distribution is strongly dependent on the size of the catalyst pores. Catalysts containing mainly mesopores, such as Sn-MCM-41 or Sn-SBA-15, prefer the production of the more bulky MMHB, whereas microporous catalysts such as Sn-β or Sn-MFI favor the production of MVG. This effect can be further enhanced by increasing the reaction temperature. At 363 K, only 20% MVG is attained using Sn-β, but at 433 K, this increases to 50%. Using a kinetic analysis, it was found that, in microporous catalysts, steric hindrance near the Sn active site in the catalyst pores plays a dominant role in favoring the reaction pathway toward MVG. Moreover, the selectivity toward both products is kinetically controlled.
Shape-selective zeolite catalysis for bioplastics production 03-07-2015
Zeolites can help synthesize cheaper plastic precursors from biologically sourced feedstocks. Producing sustainable plastics must compete with more cost-effective petrochemical-based synthesis routes. Dusselier et al. developed a zeolite-based strategy to catalyze the transformation of microbially produced lactic acid into lactide, a difficult-to-synthesize precursor of biodegradable polylactic acid plastics. The selectivity of nearly 80% is based on active site spatial confinement in the zeolite micropores. This step substantially simplifies current high-cost synthesis routes and generates nearly zero waste using current reactor technologies.
Widely picked up in non-specialized media, e.g. C&EN, Synfacts, ...
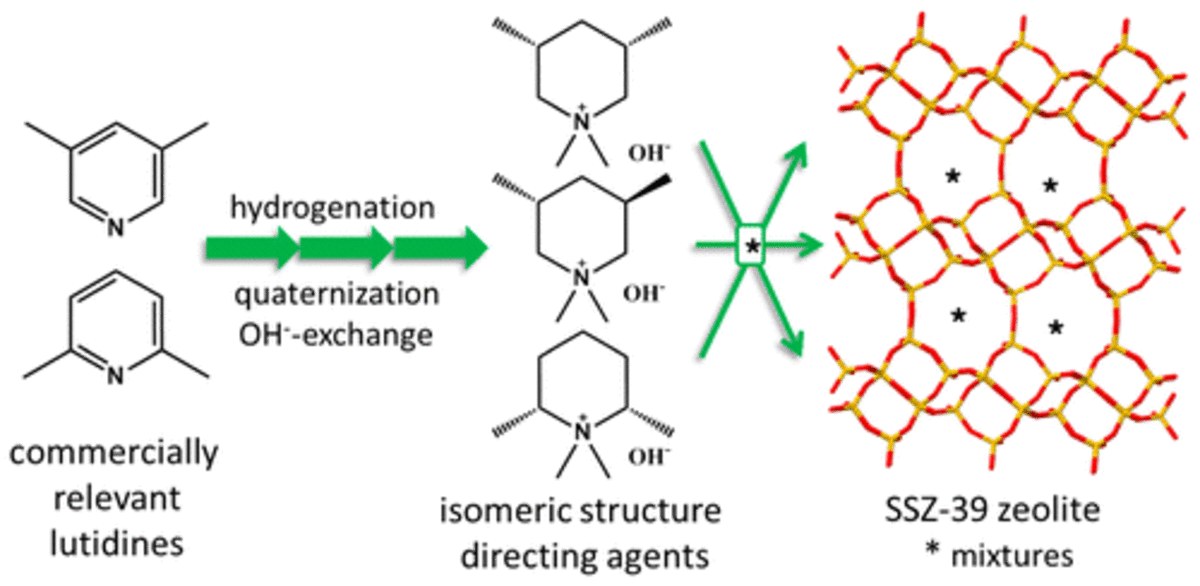
Influence of Organic Structure Directing Agent Isomer Distribution on the Synthesis of SSZ-39 30-03-2015
The aluminosilicate molecular sieve with the AEI framework topology (SSZ-39) is currently of great interest for use in a number of important applications such as exhaust gas NOx reduction and the methanol-to-olefins reaction. It is likely that advances in the synthesis of this molecular sieve will be needed for applications to proceed. Here, dimethylpiperidine based organic structure directing agents (OSDAs) are used to prepare SSZ-39, and the influence of diastereo- and structural isomeric mixtures on the synthesis of SSZ-39 is reported. Although differences in the rates of molecular sieve formation as well as preferential isomer incorporation occur, the synthesis of SSZ-39 is possible over a wide range of isomeric mixtures. These findings demonstrate that the synthesis of SSZ-39 can be accomplished with OSDA isomer mixtures that naturally occur from the synthesis of the organic precursors used to prepare the OSDAs.
Cooperative Catalysis for Multistep Biomass Conversion with Sn/Al Beta Zeolite 01-01-2015
Lewis acid Snβ-type zeolites with varying amounts of Brønsted acid Al in the framework were synthesized using a simple two-step procedure comprising partial dealumination of β zeolite under action of acid, followed by grafting with SnCl4·5H2O in dry isopropanol. Characterization of the thus-prepared Al-containing Snβ (Sn/pDeAlβ) zeolites with ICP, (pyridine probed) FTIR, and 27Al MAS NMR demonstrates the presence of Brønsted acid framework AlIII. Tetrahedral Lewis acidic SnIV is present, as ascertained by a combination of techniques such as EPMA, 119Sn Möβbauer, XPS, (pyridine probed) FTIR, and UV–vis. A closed SnIV configuration was implied by comparing of 119Sn solid-state MAS NMR and deuterated acetonitrile probed FTIR spectra with literature. The catalytic activity of the Al-containing Snβ was tested for the conversion of 1,3-dihydroxyacetone (DHA) into ethyl lactate (ELA), proceeding via pyruvic aldehyde (PAL). Despite the difference in synthesis between the classic hydrothermal Snβ reference and Sn/pDeAlβ, the activity of Sn for the Lewis acid-catalyzed hydride shift of PAL to ELA is similar. Yet, the overall reaction rate of DHA into ELA is faster with Sn/pDeAlβ because Brønsted acidity of the remaining framework AlIIIfacilitates the rate-determining dehydration of DHA into PAL. Materials containing moderate amounts of Al (0.3 wt % Al) show the highest ELA productivities, leading to a record value of 2113 g ELA·kg catalyst–1·h–1 at 363 K. The cooperative effect of Lewis SnIV and Brønsted AlIII acid sites is verified by comparing catalytic data with physical mixtures of partially dealuminated β zeolite and Al-free Snβ.
2014
Direct Catalytic Conversion of Cellulose to Liquid Straight-Chain Alkanes 09-09-2014
High yields of liquid straight-chain alkanes were obtained directly from cellulosic feedstock in a one-pot biphasic catalytic system. The catalytic reaction proceeds at elevated temperatures under hydrogen pressure in the presence of tungstosilicic acid, dissolved in the aqueous phase, and modified Ru/C, suspended in the organic phase. Tungstosilicic acid is primarily responsible for cellulose hydrolysis and dehydration steps, while the modified Ru/C selectively hydrogenates intermediates en route to the liquid alkanes. Under optimal conditions, microcrystalline cellulose is converted to 82% n-decane-soluble products, mainly n-hexane, within a few hours, with a minimum formation of gaseous and char products. The dominant route to the liquid alkanes proceeds via 5-hydroxymethylfurfural (HMF), whereas the more common pathway via sorbitol appears to be less efficient. High liquid alkane yields were possible through (i) selective conversion of cellulose to glucose and further to HMF by gradually heating the reactor, (ii) a proper hydrothermal modification of commercial Ru/C to tune its chemoselectivity to furan hydrogenation rather than glucose hydrogenation, and (iii) the use of a biphasic reaction system with optimal partitioning of the intermediates and catalytic reactions. The catalytic system is capable of converting subsequent batches of fresh cellulose, enabling accumulation of the liquid alkanes in the organic phase during subsequent runs. Its robustness is illustrated in the conversion of the raw (soft)wood sawdust.
In the top 29 of most accessed EES papers in 2015
Will Zeolite-Based Catalysis be as Relevant in Future Biorefineries as in Crude Oil Refineries? 25-07-2014
Transition from petroleum- to biomass-based fuel economy will require new conversion strategies. In a petroleum refinery, particular hydrocarbon fractions from crude oil are catalytically converted into high-grade fuels. Certain zeolite catalysts are performing exceptionally well. Unlike petroleum fractions, biomass-derived compounds have a high oxygen content requiring low-temperature catalytic aqueous phase processes for selective conversion and stability of zeolite catalysts in hot liquid water. It will be shown that recent developments in zeolite synthesis and modification allow adapting zeolite properties to achieve selective conversion of biomass compounds/fractions as well.
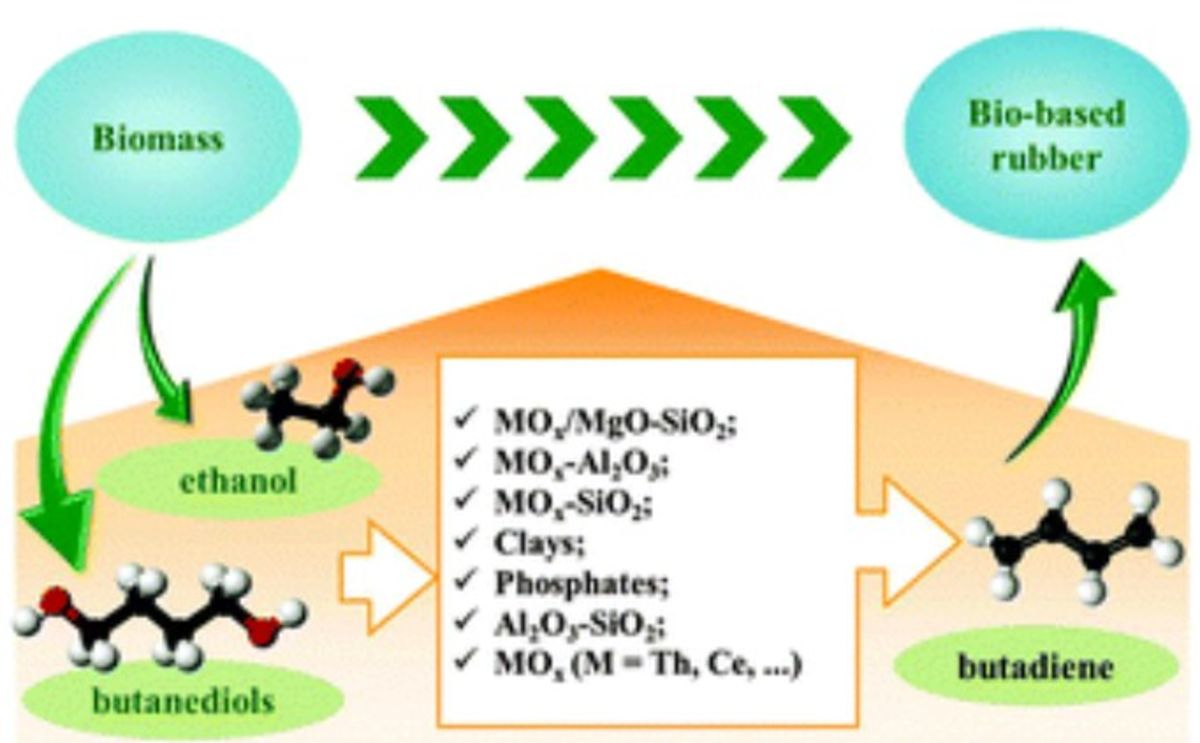
Review of old chemistry and new catalytic advances in the on-purpose synthesis of butadiene. 04-07-2014
Increasing demand for renewable feedstock-based chemicals is driving the interest of both academic and industrial research to substitute petrochemicals with renewable chemicals from biomass-derived resources. The search towards novel platform chemicals is challenging and rewarding, but the main research activities are concentrated on finding efficient pathways to produce familiar drop-in chemicals and polymer building blocks. A diversity of industrially important monomers like alkenes, conjugated dienes, unsaturated carboxylic acids and aromatic compounds are thus targeted from renewable feedstock. In this context, on-purpose production of 1,3-butadiene from biomass-derived feedstock is an interesting example as its production is under pressure by uncertainty of the conventional fossil feedstock. Ethanol, obtained via fermentation or (biomass-generated) syngas, can be converted to butadiene, although there is no large commercial activity today. Though practised on a large scale in the beginning of the 20th century, there is a growing worldwide renewed interest in the butadiene-from-ethanol route. An alternative route to produce butadiene from biomass is through direct carbohydrate and gas fermentation or indirectly via the dehydration of butanediols. This review starts with a brief discussion on the different feedstock possibilities to produce butadiene, followed by a comprehensive summary of the current state of knowledge regarding advances and achievements in the field of the chemocatalytic conversion of ethanol and butanediols to butadiene, including thermodynamics and kinetic aspects of the reactions with discussions on the reaction pathways and the type of catalysts developed.
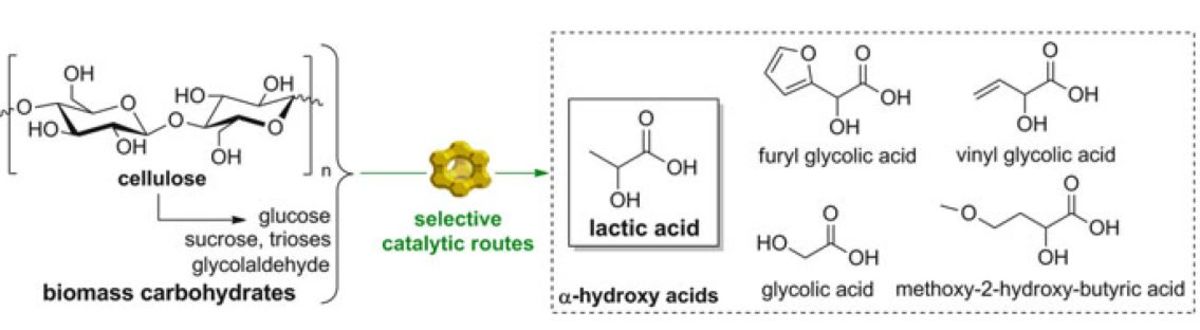
Selective catalysis for cellulose conversion to lactic acid and other α-hydroxy acids 19-03-2014
This review discusses topical chemical routes and their catalysis for the conversion of cellulose, hexoses, and smaller carbohydrates to lactic acid and other useful α-hydroxy acids. Lactic acid is a top chemical opportunity from carbohydrate biomass as it not only features tremendous potential as a chemical platform molecule; it is also a common building block for commercially employed green solvents and near-commodity bio-plastics. Its current scale fermentative synthesis is sufficient, but it could be considered a bottleneck for a million ton scale breakthrough. Alternative chemical routes are therefore investigated using multifunctional, often heterogeneous, catalysis. Rather than summarizing yields and conditions, this review attempts to guide the reader through the complex reaction networks encountered when synthetic lactates from carbohydrate biomass are targeted. Detailed inspection of the cascade of reactions emphasizes the need for a selective retro-aldol activity in the catalyst. Recently unveiled catalytic routes towards other promising α-hydroxy acids such as glycolic acid, and vinyl and furyl glycolic acids are highlighted as well.
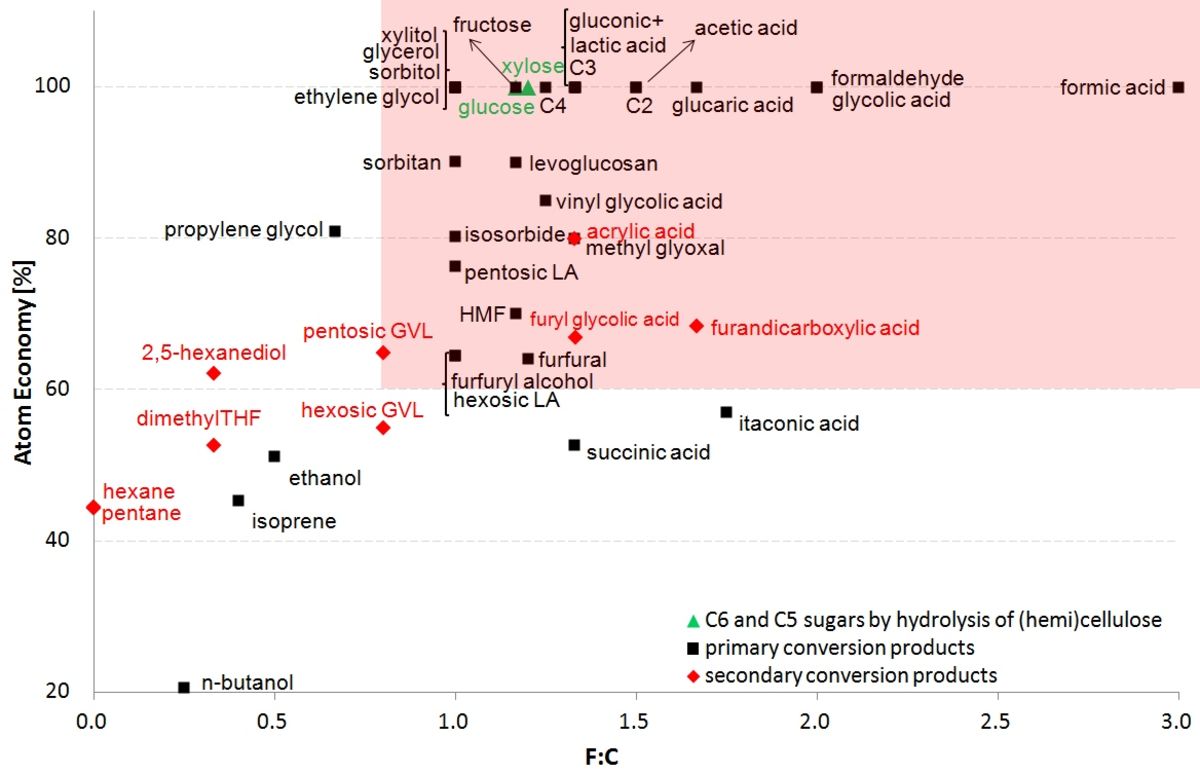
Top Chemical Opportunities from Carbohydrate Biomass: A Chemist’s View of the Biorefinery 14-03-2014
To guide the choice of renewable chemicals from carbohydrate biomass and their production, this review adopts the original van Krevelen plots and develops alternative diagrams by introducing a functionality parameter F and a functionality index F:C (rather than O:C). This index is more powerful than the O index to describe the importance of functional groups. Such plots are ideal to assess the effect of several reaction types on the overall functionality in biomass conversion. The atom economy is an additional arbitrator in the evaluation of the reaction types. The assessment is illustrated in detail for the case of carbohydrate resources, and about 25 chemicals, including drop-ins as well as novel chemicals, are selected.
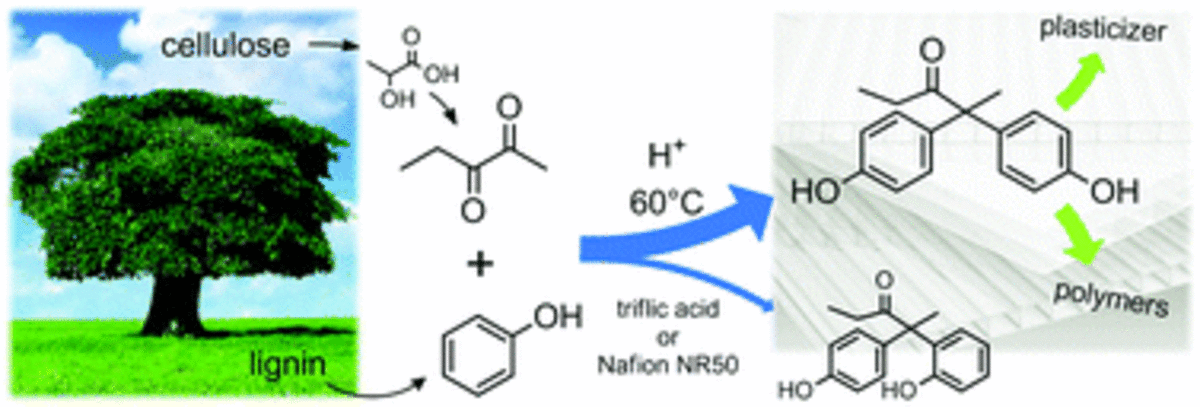
Regioselective synthesis of renewable bisphenols from 2,3-pentanedione and their application as plasticizers 24-02-2014
2,3-Pentanedione (2,3-PD), a bio-based chemical derived from lactic acid, has the potential to serve as a precursor for the synthesis of novel bisphenols. We developed a solvent-free catalytic strategy for the condensation of phenol with 2,3-PD by using acid catalysts at temperatures ranging from 323 to 373 K. Various soluble and solid acids exhibit high activity, while a high chemoselectivity to bisphenol requires a high phenol to 2,3-PD molar ratio. Bisphenol yields as high as 84% are for instance reported in an excess of phenol in the presence of Nafion NR50. Recycling of the Nafion catalyst after washing with ethanol at room temperature is demonstrated. The regioselectivity in the bisphenol fraction is influenced by the acid strength. A clear trend is presented in which the regioselectivity towards the desired p,p′-isomers increases with increasing acid strength, showing p,p′/o,p′-isomer ratios as high as 100. A tentative mechanism is discussed based on the ionic versusnon-ionic pathway. The purified 2,3-PD-derived p,p′-bisphenols are assessed as plasticizers for polyethylene terephthalate (PET), showing promising properties similar to that of the reference bisphenol A, but with a broader processing window due to the lower melting point and higher thermal stability under an inert atmosphere.
Induced Chirality in a Metal–Organic Framework by Postsynthetic Modification for Highly Selective Asymmetric Aldol Reactions 20-02-2014
2013
Conversion of Sugars to Ethylene Glycol with Nickel Tungsten Carbide in a Fed-Batch Reactor: High Productivity and Reaction Network Elucidation 15-10-2013
Bifunctional nickel tungsten carbide catalysis was used for the conversion of aqueous sugar solutions into short-chain polyols such as ethylene glycol. It is shown that very concentrated sugar solutions, viz. up to 0.2 kg L−1, can be converted without loss of ethylene glycol selectivity by gradually feeding the sugar solution. Detailed investigation of the reaction network shows that, under the applied reaction conditions, glucose is converted viaa retro-aldol reaction into glycol aldehyde, which is further transformed into ethylene glycol by hydrogenation. The main byproducts are sorbitol, erythritol, glycerol and 1,2-propanediol. They are formed through a series of unwanted side reactions including hydrogenation, isomerisation, hydrogenolysis and dehydration. Hydrogenolysis of sorbitol is only a minor source of ethylene glycol. To assess the relevance of the fed-batch system in biomass conversions, both the influence of the catalyst composition and the reactor setup parameters like temperature, pressure and glucose addition rate were optimized, culminating in ethylene glycol yields up to 66% and separately, volume productivities of nearly 300 gEG L−1 h−1.
Bridging racemic lactate esters with stereoselective polylactic acid using commercial lipase catalysis 14-08-2013
A productive and enantioselective hydrolysis of racemic mixtures of lactate esters with commercial Candida rugosa lipase was performed. This step contributes to a novel envisioned route for stereoselective PLA production by combining recent chemocatalytic developments with this biocatalytic contribution, foreseeing two separate l- and d-lactate enantiomer streams. A study of the hydrolysis kinetics identified an unexpected rate determining step at the origin of an unprecedented ester reactivity order.
Productive sugar isomerization with highly active Sn in dealuminated beta zeolites 02-08-2013
A water-tolerant Lewis acid catalyst was synthesized by grafting SnIV in isopropanol under reflux onto dealuminated zeolites with the BEA ([small beta]) topology. This synthesis method allows the production of highly active Sn[small beta]-type catalysts without the need for long hydrothermal syntheses or hydrogen fluoride, while using cheap Sn-precursors, industrially available [small beta] zeolites and standard catalyst synthesis unit operations. Extensive characterization of the best catalyst shows highly dispersed Sn in the zeolite matrix (XRD, 29Si MAS NMR and 1H MAS NMR) without the formation of SnO2 (XRD and UV-Vis). The catalyst was tested for the model isomerization of sugars such as glucose to fructose. The catalytic activity proved to be purely heterogeneous and the catalyst was recycled and reused without significant loss in activity. Isomerization productivities above 4 kg product per kg of catalyst per hour are reported with appreciably low Sn loadings, corresponding to exceptionally high turnover frequencies, viz. 500 cycles per Sn per hour at 110 [degree]C, which surpass the activity per Sn of the original hydrothermally synthesized Sn-Beta.
Toward Functional Polyester Building Blocks from Renewable Glycolaldehyde with Sn Cascade Catalysis 25-06-2013
Having been inspired by formose-based hypotheses surrounding the origin of life, we report on a novel catalytic route toward a series of recently discovered four-carbon α-hydroxy acids (AHA) and their esters from accessible and renewable glycolaldehyde (GA) in various solvents. The synthesis route follows a cascade type reaction network, and its mechanism with identification of the rate-determining step was investigated with in situ 13C NMR. The mechanistic understanding led to optimized reaction conditions with higher overall rates of AHA formation by balancing Brønsted and Lewis acid activity, both originating from the tin halide catalyst. An optimal H+/Sn ratio of 3 was identified, and this number was surprisingly irrespective of the Sn oxidation state. Further rate enhancement was accomplished by adding small amounts of water to the reaction mixture, boosting the rate by a factor of 4.5 compared with pure methanol solvent. The cascade reaction selectively yields near 60% methyl-4-methoxy-2-hydroxybutanoate (MMHB). In the optimized rate regime in methanol, an initial TOF of 7.4 molGA molSn–1 h–1 was found. In sterically hindered alcohols (isopropyl alcohol), the rate of AHA formation was even higher, and the corresponding vinyl glycolate esters arose as the main product. Vinyl glycolic acid, 2,4-dihydroxybutanoic acid, and its lactone were formed significantly in nonprotic solvent. The corresponding AHAs have serious potential as building blocks in novel biobased polymers with tunable functionality. The incorporation of vinyl glycolic acid in polylactic acid-based polyesters is illustrated, and postmodification at the vinyl side groups indeed allows access to a range of properties, such as tunable hydrophilicity, which is otherwise difficult to attain for pure poly(l-lactic acid).
Tailoring nanohybrids and nanocomposites for catalytic applications 02-04-2013
Research on and development of inorganic-organic nanohybrids and nanocomposite materials has attracted increasing attention in recent years. Synthetic strategies for such materials vary from grafting or co-condensation of Si and C sources to the impregnation of silica with polymers. Nanohybrids, prepared using organosilanes, and nanocomposites, obtained by hard or soft templated synthesis, are discussed. Various strategies will be presented that demonstrate how additional carbon properties can be exploited maximising the activity, selectivity and stability of composite materials as solid catalysts. Composite materials allow for the extensive engineering of a catalyst enabling careful tuning of the type, amount and position of active sites, as well as the porosity and hydrophilic nature of the final catalyst. These materials not only combine the advantages of silica (e.g. thermal stability, rigidity, ordering) and carbon (e.g. flexibility, ductility) but also allow their synergetic action in various catalytic applications.
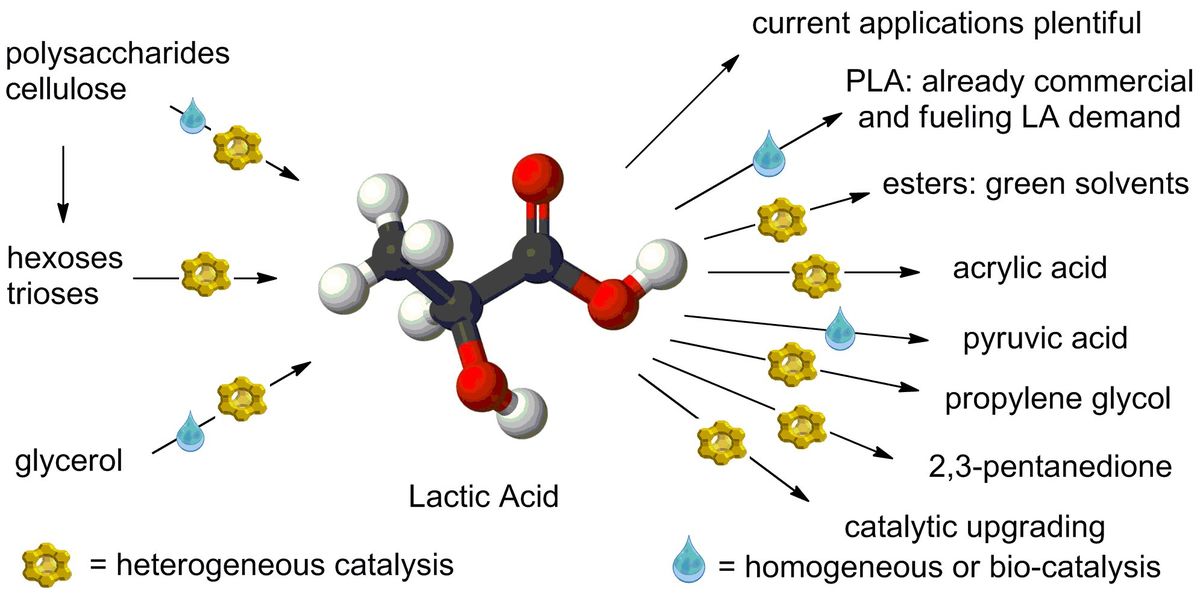
Lactic acid as a platform chemical in the biobased economy: the role of chemocatalysis. 06-03-2013
Upcoming bio-refineries will be at the heart of the manufacture of future transportation fuels, chemicals and materials. A narrow number of platform molecules are envisioned to bridge nature's abundant polysaccharidefeedstock to the production of added-value chemicals and intermediate building blocks. Such platform molecules are well-chosen to lie at the base of a large product assortment, while their formation should be straightforward from the refined biomass, practical and energy efficient, without unnecessary loss of carbon atoms. Lactic acid has been identified as one such high potential platform. Despite its established fermentationroute, sustainability issues – like gypsum waste and cost factors due to multi-step purification and separation requirements – will arise as soon as the necessary orders of magnitude larger volumes are needed. Innovative production routes to lactic acid and its esters are therefore under development, converting sugars and glycerol in the presence of chemocatalysts. Moreover, catalysis is one of the fundamental routes to convert lactic acid into a range of useful chemicals in a platform approach. This contribution attempts a critical overview of all advances in the field of homogeneous and heterogeneous catalysis and recognises a great potential of some of these chemocatalytic approaches to produce and transform lactic acid as well as some other promising α-hydroxy acids.
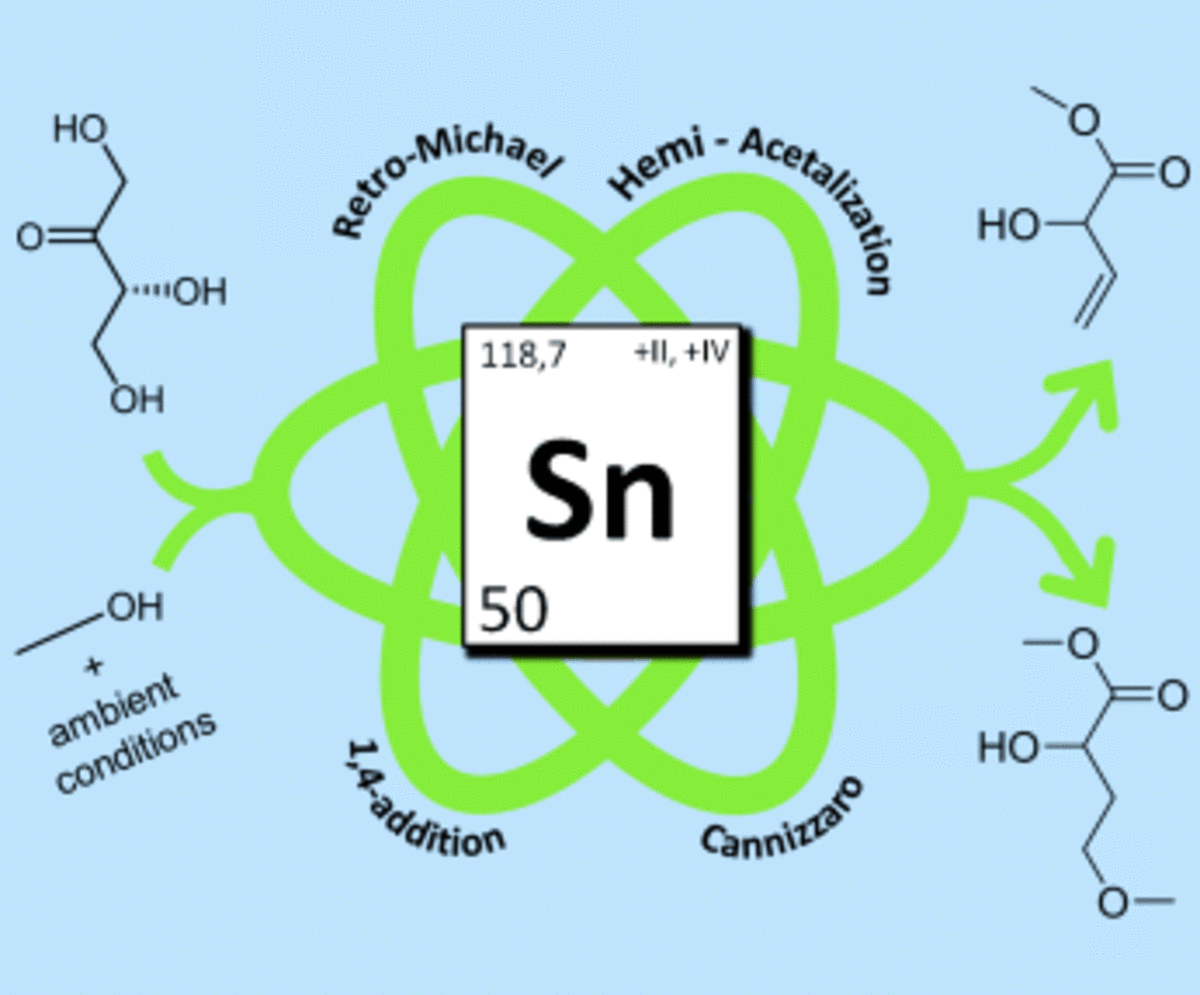
Mechanistic Insight into the Conversion of Tetrose Sugars to Novel α-Hydroxy Acid Platform Molecules. 01-01-2013
α-Hydroxy acids (AHAs) such as lactic acid are considered platform molecules in the biorefinery concept and have high-end applications in solvents and biodegradable polyester plastics. The synthesis of AHAs with a four-carbon backbone structure is a recently emerging field. New biomass-related routes towards their production could stimulate their practical use in new polyester plastics. Herein, we report the unique catalytic activity of soluble tin metal salts for converting tetroses, namely erythrulose and erythrose, into new four-carbon-backbone AHAs such as methyl vinylglycolate and methyl-4-methoxy-2-hydroxybutanoate. An in situ NMR study together with deuterium labeling experiments and control experiments with intermediates allowed us to propose a detailed reaction pathway.
2012
Lipoteichoic acid is an important microbe-associated molecular pattern of Lactobacillus rhamnosus GG 15-12-2012
Probiotic bacteria are increasingly used as immunomodulatory agents. Yet detailed molecular knowledge on the immunomodulatory molecules of these bacteria is lagging behind. Lipoteichoic acid (LTA) is considered a major microbe-associated molecular pattern (MAMP) of Gram-positive bacteria. However, many details and quantitative data on its immune signalling capacity are still unknown, especially in beneficial bacteria. Recently, we have demonstrated that a dltD mutant of the model probiotic Lactobacillus rhamnosus GG (LGG), having modified LTA molecules, has an enhanced probiotic efficacy in a DSS-induced colitis model as compared to wild-type.

Tuning the Acid/Metal Balance of Carbon Nanofiber-Supported Nickel Catalysts for Hydrolytic Hydrogenation of Cellulose 06-12-2012
Carbon nanofibers (CNFs) are a class of graphitic support materials with considerable potential for catalytic conversion of biomass. Earlier, we demonstrated the hydrolytic hydrogenation of cellulose over reshaped nickel particles attached at the tip of CNFs. The aim of this follow‐up study was to find a relationship between the acid/metal balance of the Ni/CNFs and their performance in the catalytic conversion of cellulose. After oxidation and incipient wetness impregnation with Ni, the Ni/CNFs were characterized by various analytical methods. To prepare a selective Ni/CNF catalyst, the influences of the nature of oxidation agent, Ni activation, and Ni loading were investigated. Under the applied reaction conditions, the best result, that is, 76 % yield in hexitols with 69 % sorbitol selectivity at 93 % conversion of cellulose, was obtained on a 7.5 wt % Ni/CNF catalyst prepared by chemical vapor deposition of CH4 on a Ni/γ‐Al2O3 catalyst, followed by oxidation in HNO3 (twice for 1 h at 383 K), incipient wetness impregnation, and reduction at 773 K under H2. This preparation method leads to a properly balanced Ni/CNF catalyst in terms of Ni dispersion and hydrogenation capacity on the one hand, and the number of acidic surface‐oxygen groups responsible for the acid‐catalyzed hydrolysis on the other.
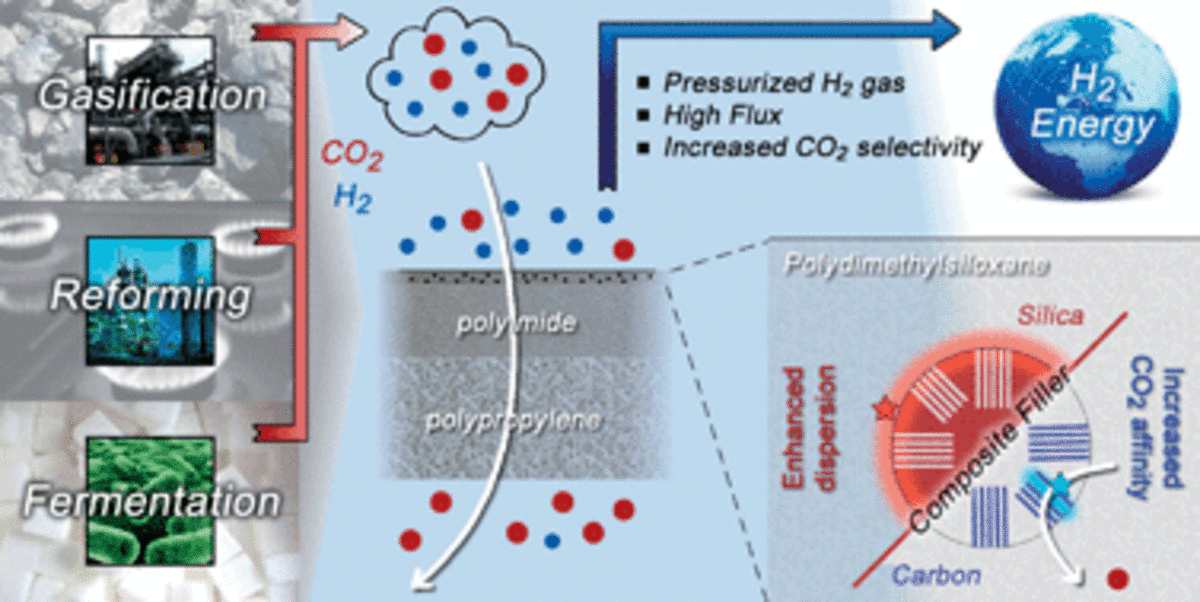
CO2 reverse selective mixed matrix membranes for H2 purification by incorporation of carbon-silica fillers 14-10-2012
By filling a PDMS top layer with porous carbon-silica microspheres, a defect-free mixed matrix membrane was created with notable CO2 reverse selective separation properties. For the separation of CO2 over H2 at room temperature and 10 bar inlet pressure, these membranes demonstrate high CO2 gas fluxes up to 3 [times] 10-7 mol cm-2 s-1, in combination with ideal separation factors in the range of 6 to 9. The present separation data signify an important step forward in the removal of CO2 from H2 using a reverse selective separation strategy. Moreover, they elucidate the potential of such mixed matrix membranes in the emerging field of CO2 separation.
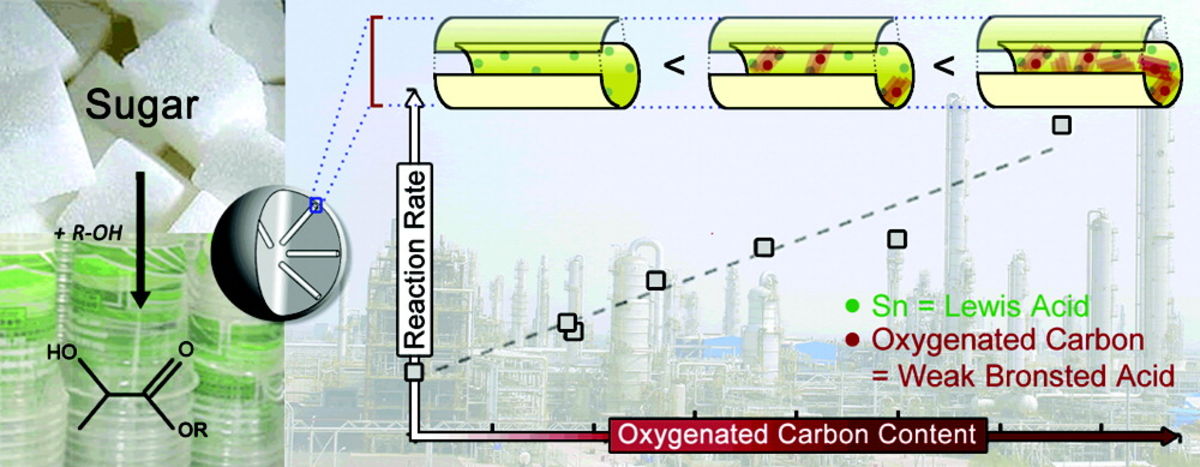
Fast and Selective Sugar Conversion to Alkyl Lactate and Lactic Acid with Bifunctional Carbon–Silica Catalysts 02-05-2012
A novel catalyst design for the conversion of mono- and disaccharides to lactic acid and its alkyl esters was developed. The design uses a mesoporous silica, here represented by MCM-41, which is filled with a polyaromatic to graphite-like carbon network. The particular structure of the carbon-silica composite allows the accommodation of a broad variety of catalytically active functions, useful to attain cascade reactions, in a readily tunable pore texture. The significance of a joint action of Lewis and weak Brønsted acid sites was studied here to realize fast and selective sugar conversion. Lewis acidity is provided by grafting the silica component with Sn(IV), while weak Brønsted acidity originates from oxygen-containing functional groups in the carbon part. The weak Brønsted acid content was varied by changing the amount of carbon loading, the pyrolysis temperature, and the post-treatment procedure. As both catalytic functions can be tuned independently, their individual role and optimal balance can be searched for. It was thus demonstrated for the first time that the presence of weak Brønsted acid sites is crucial in accelerating the rate-determining (dehydration) reaction, that is, the first step in the reaction network from triose to lactate. Composite catalysts with well-balanced Lewis/Brønsted acidity are able to convert the trioses, glyceraldehyde and dihydroxyacetone, quantitatively into ethyl lactate in ethanol with an order of magnitude higher reaction rate when compared to the Sn grafted MCM-41 reference catalyst. Interestingly, the ability to tailor the pore architecture further allows the synthesis of a variety of amphiphilic alkyl lactates from trioses and long chain alcohols in moderate to high yields. Finally, direct lactate formation from hexoses, glucose and fructose, and disaccharides composed thereof, sucrose, was also attempted. For instance, conversion of sucrose with the bifunctional composite catalyst yields 45% methyl lactate in methanol at slightly elevated reaction temperature. The hybrid catalyst proved to be recyclable in various successive runs when used in alcohol solvent.
2010
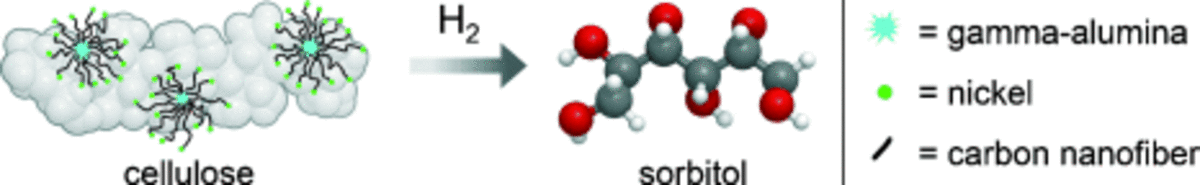
Selective Bifunctional Catalytic Conversion of Cellulose over Reshaped Ni Particles at the Tip of Carbon Nanofibers 02-05-2010
Carbon nanofibers grown over Ni supported on γ‐Al2O3 act as efficient catalysts for the one‐pot conversion of cellulose to sugar alcohols, owing to the enhanced accessibility of the water‐insoluble substrate towards the active catalytic sites. The new catalyst design concept yields unprecedented results for selective cellulose conversion using inexpensive Ni catalysts.